Open-Label, Single-Center, Randomized, 2-way Crossover Study Evaluating Relative Bioavailability of OC-01 (Varenicline) Nasal Spray as Compared to Orally Administered Varenicline (the ZEN Study)
| Molecule | Varenicline solution nasal spray (formerly known as OC-01) |
| Therapeutic focus | Dry eye disease2 |
| Phase | 1 |
| Status | Completed |
| Participants | 22 |
| Completion Date | November 15, 2019 |
Note: This clinical trial was not included in the submission which resulted in Tyrvaya® approval.
1. https://clinicaltrials.gov/study/NCT04072146; 2. Nau J. et al. Clinical Therapeutics. 2021;43:1595–1607.

Trial Design1
The study objective was to compare the systemic bioavailability of varenicline nasal spray with that of varenicline administered orally1
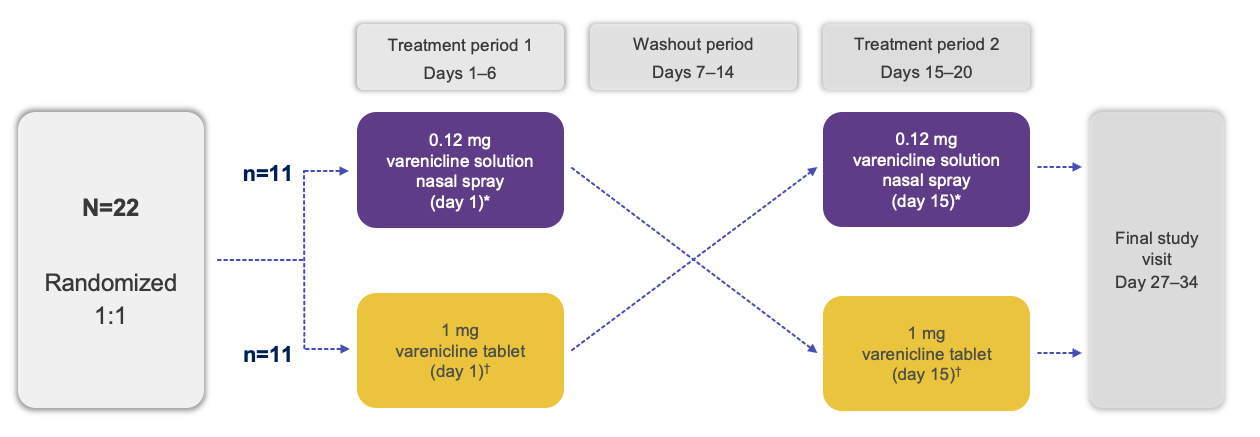
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 *Delivered as one 50-μL spray of varenicline 1.2 mg/mL into each nostril; †Delivered as a single oral tablet.1
OD, once daily; FDA, Food and Drug Administration.
1. Nau J. et al. Clinical Therapeutics. 2021;43:1595–607; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
OD, once daily; FDA, Food and Drug Administration.
1. Nau J. et al. Clinical Therapeutics. 2021;43:1595–607; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Baseline Characteristics
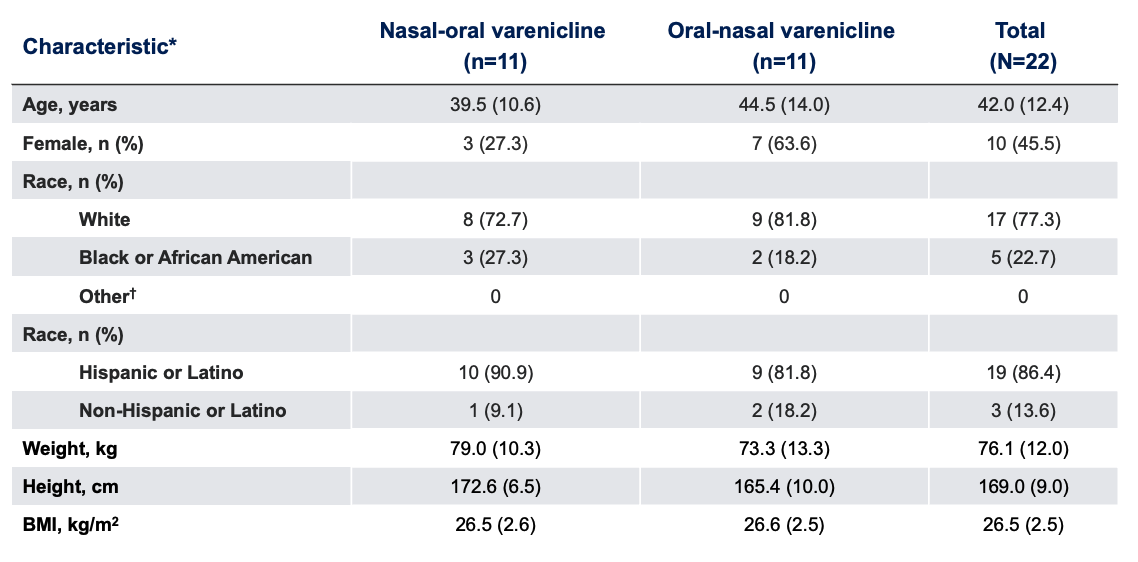
*Data presented as mean (SD), unless otherwise specified.
BMI, body mass index.
Nau J. et al.Clinical Therapeutics. 2021;43:1595–1607.
BMI, body mass index.
Nau J. et al.Clinical Therapeutics. 2021;43:1595–1607.
Efficacy Outcome Measures – Primary Endpoint
Mean change in systemic exposure over time (n=16)1
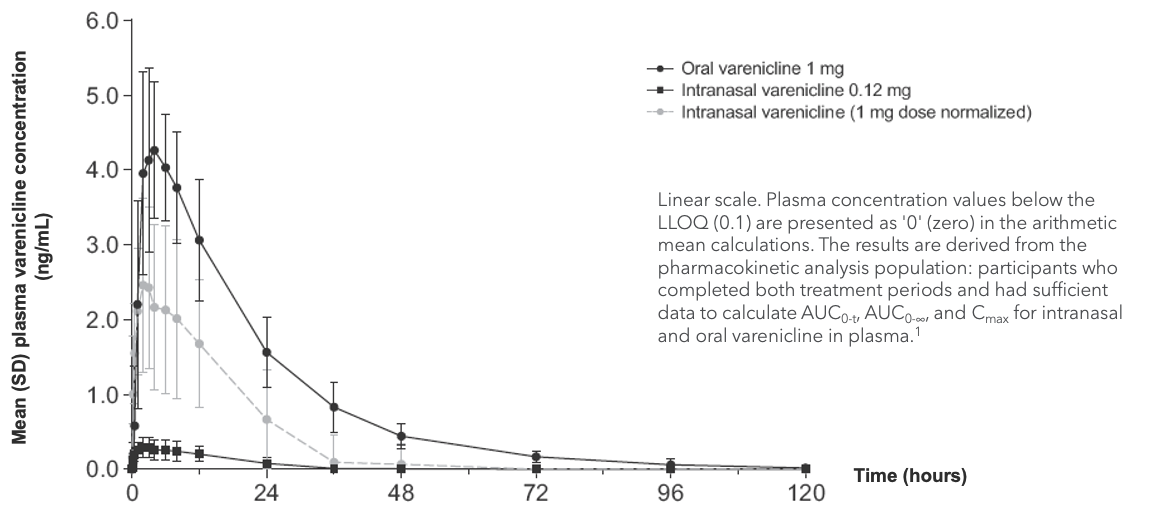
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2
AUC, area under the curve; Cmax, maximum observed concentration; LLOQ, lower limit of quantification.
1. Nau J. et al.Clinical Therapeutics. 2021;43:1595–1607; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
AUC, area under the curve; Cmax, maximum observed concentration; LLOQ, lower limit of quantification.
1. Nau J. et al.Clinical Therapeutics. 2021;43:1595–1607; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Safety Outcome Measures*1
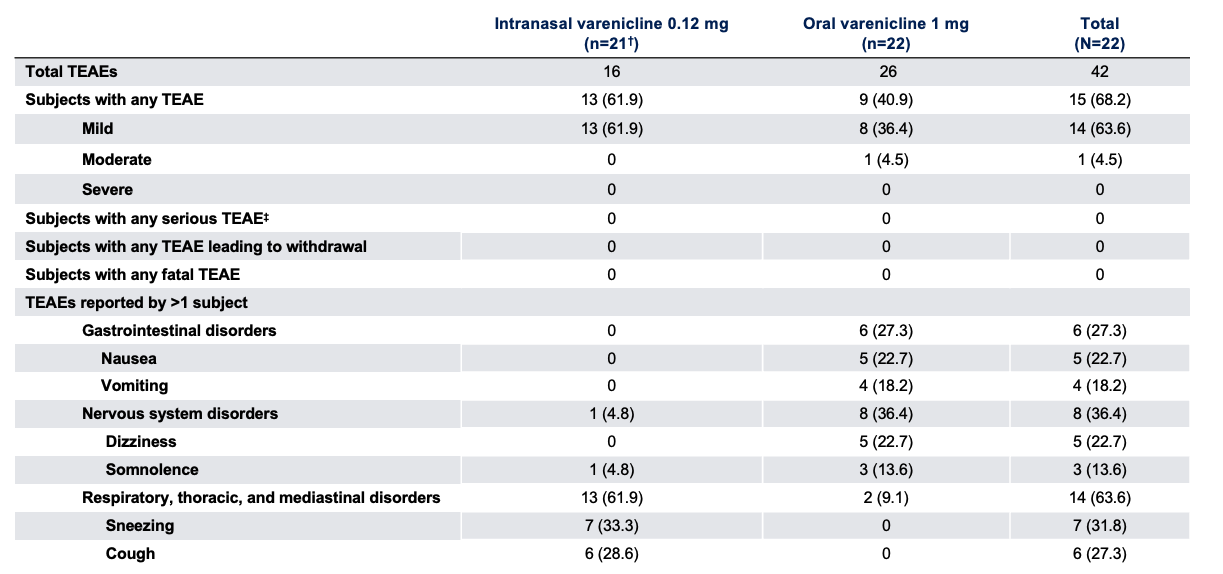
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 *Data presented as n (%); †One participant completed treatment period 1 with oral varenicline but was discontinued from the study before dosing in treatment period 2 due to adverse events unrelated to study treatment (headache and increased blood pressure); ‡Serious TEAEs were defined as those resulting in death, a life-threatening adverse event, inpatient hospitalization or prolonged hospitalization, persistent or significant incapacity or substantial disruption to normal life functions, a congenital anomaly or birth defect in an offspring, or any other medically important event that could jeopardize the participant and require medical intervention to prevent such an outcome.1
TEAE, treatment-emergent adverse event.
1. Nau J. et al.Clinical Therapeutics.2021;43:1595–1607; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
TEAE, treatment-emergent adverse event.
1. Nau J. et al.Clinical Therapeutics.2021;43:1595–1607; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.

