Matching-adjusted indirect comparison of phase 3 clinical trial outcomes: OC-01 (varenicline solution) nasal spray and cyclosporine A 0.05% ophthalmic emulsion for the treatment of dry eye disease
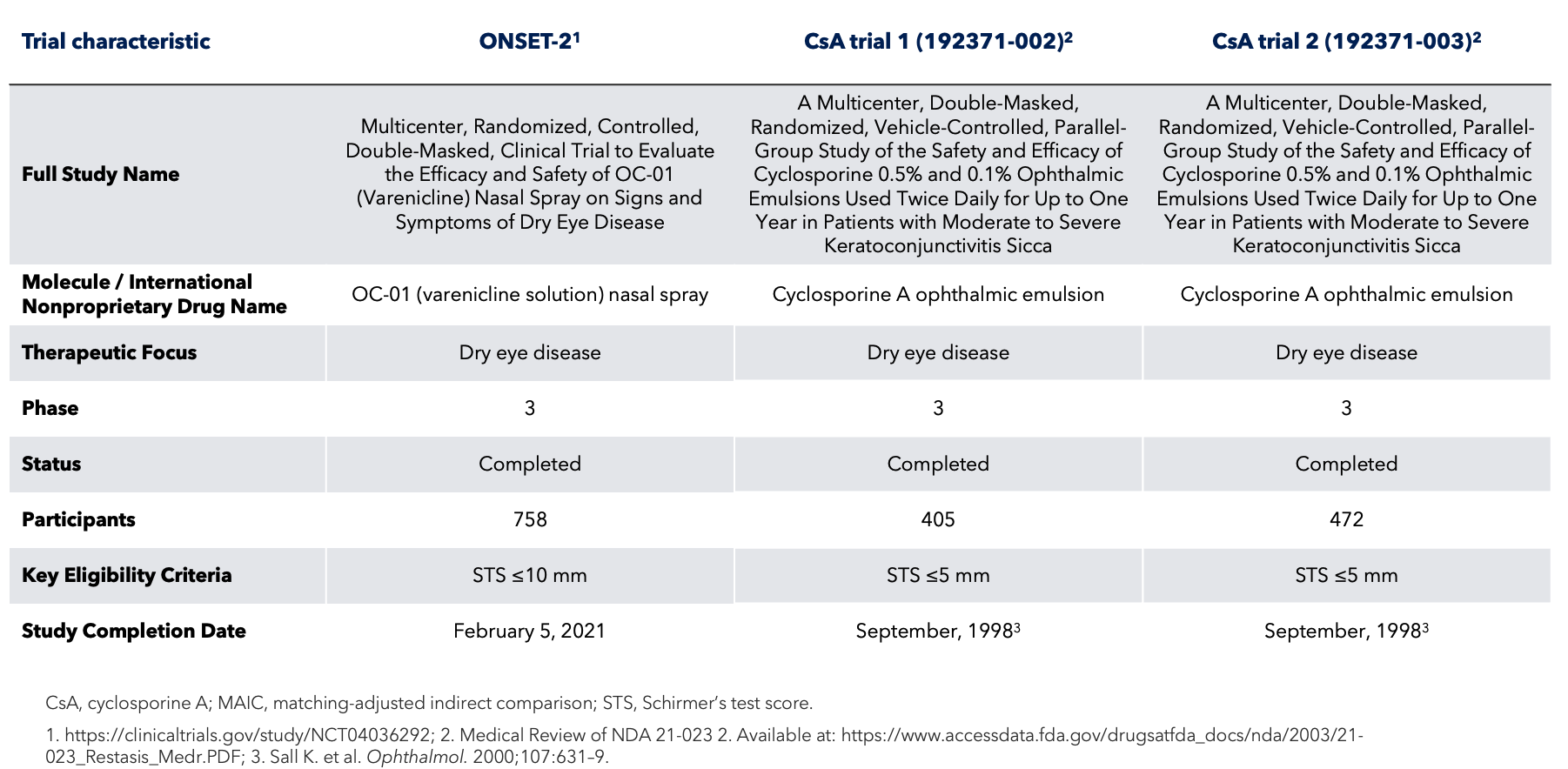

Study Design
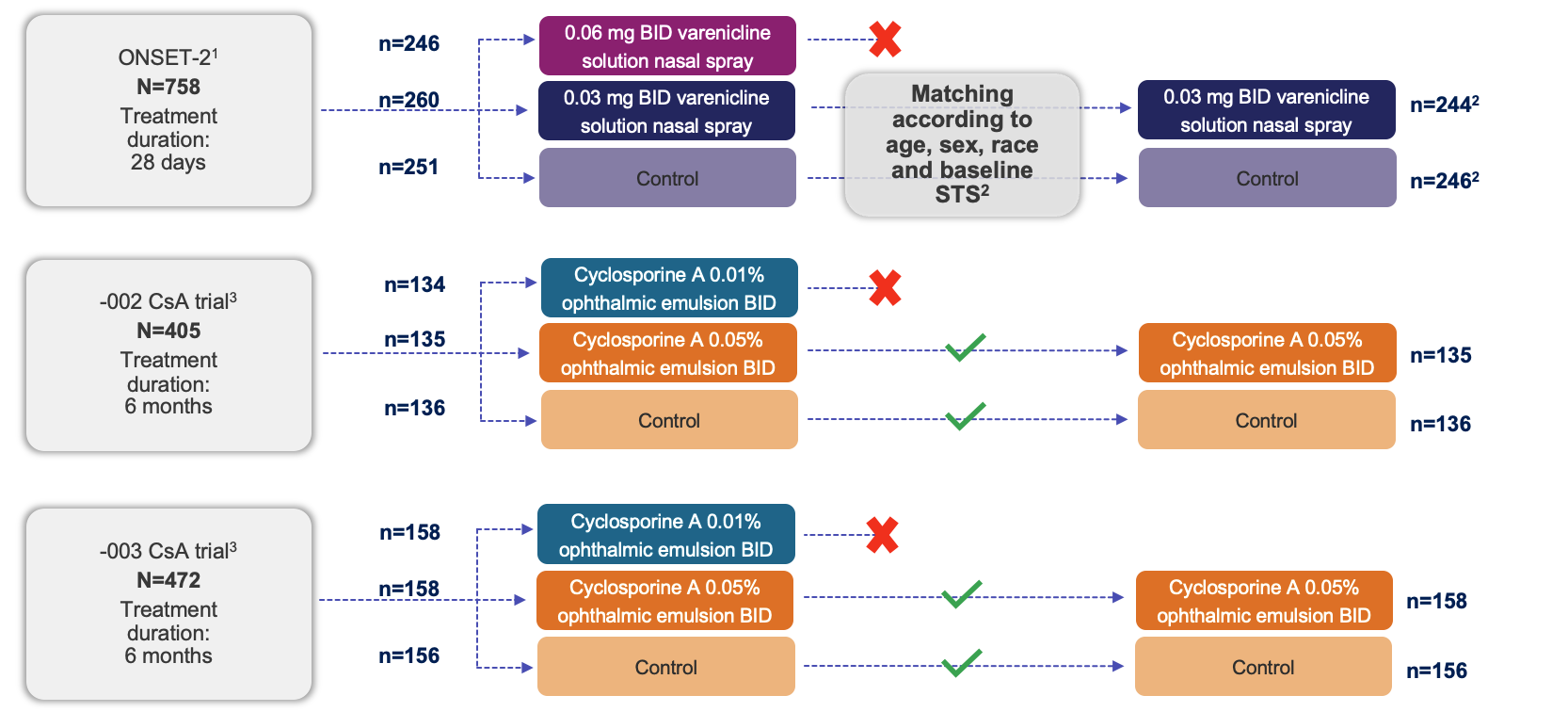
BID, twice daily; STS, Schirmer’s test score.
1. Wirta D. et al. Ophthalmology. 2022;129:379–387; 2. Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902; 3. Medical Review of NDA 21-023 2. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2003/21-023_Restasis_Medr.PDF.
1. Wirta D. et al. Ophthalmology. 2022;129:379–387; 2. Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902; 3. Medical Review of NDA 21-023 2. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2003/21-023_Restasis_Medr.PDF.
Strengths and Limitations of MAIC Design
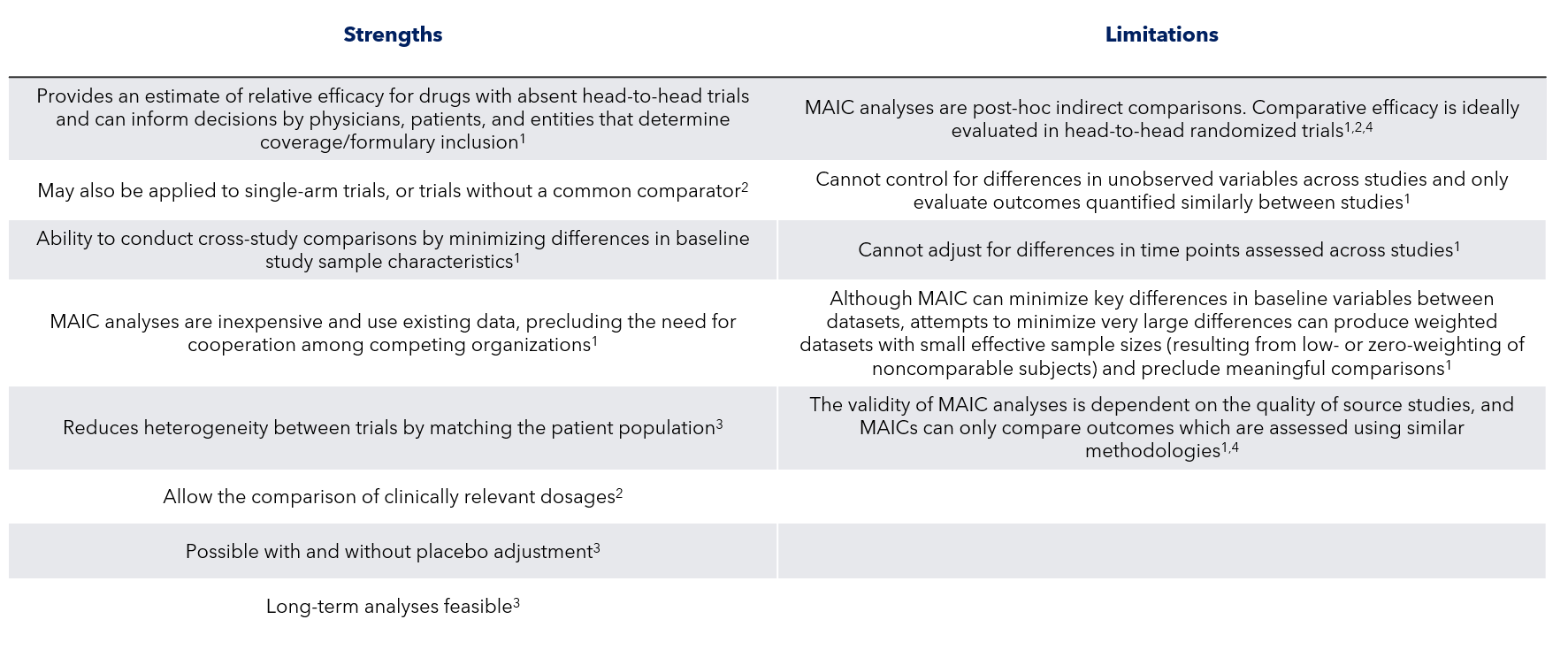
MAIC, matching-adjusted indirect comparison.
1. White D. et al. J Manag Care Spec Pharm. 2023;29:69–79; 2. Signorovitch JE. et al. Value Health. 2012;15:940–947; 3. Choy E. et al. Arthritis Res Ther. 2019;21:32; 4. Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
1. White D. et al. J Manag Care Spec Pharm. 2023;29:69–79; 2. Signorovitch JE. et al. Value Health. 2012;15:940–947; 3. Choy E. et al. Arthritis Res Ther. 2019;21:32; 4. Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
ONSET-2 vs CsA Trials Results
Baseline Characteristics With Weighting
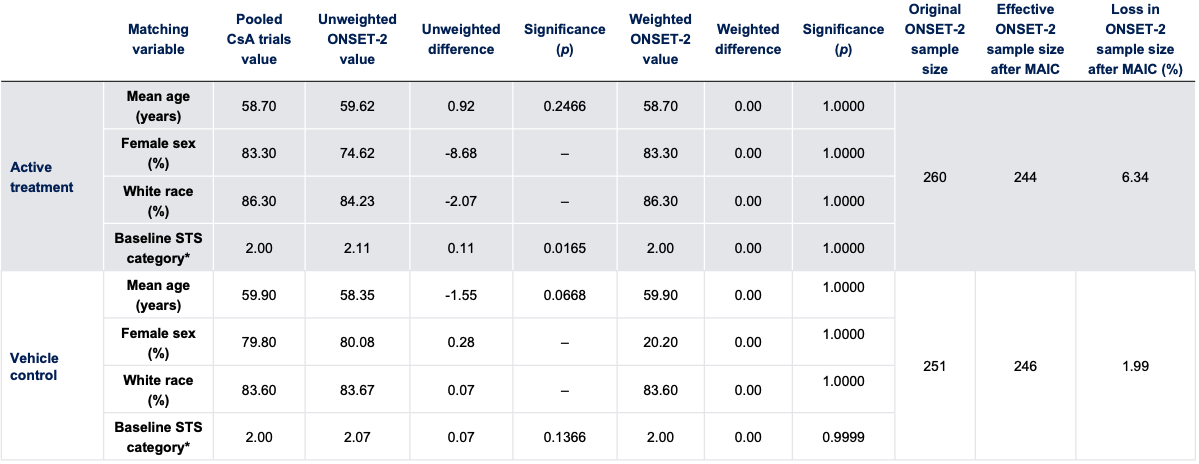
*In the CsA trials, the continuous variable STS was reduced to a categorical 5-point variable as follows: 1 (<3 mm), 2 (3–6 mm), 3 (7–10 mm), 4 (11–14 mm), and 5 (>14 mm). All ONSET-2 data were recoded to match this scale.
CsA, cyclosporine A; MAIC, matching-adjusted indirect comparison; STS, Schirmer’s test score.
Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
CsA, cyclosporine A; MAIC, matching-adjusted indirect comparison; STS, Schirmer’s test score.
Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
ONSET-2 vs CsA Trials Results
Efficacy Outcome Measures – Common Endpoint
Mean change in STS (with anesthesia) category* from baseline
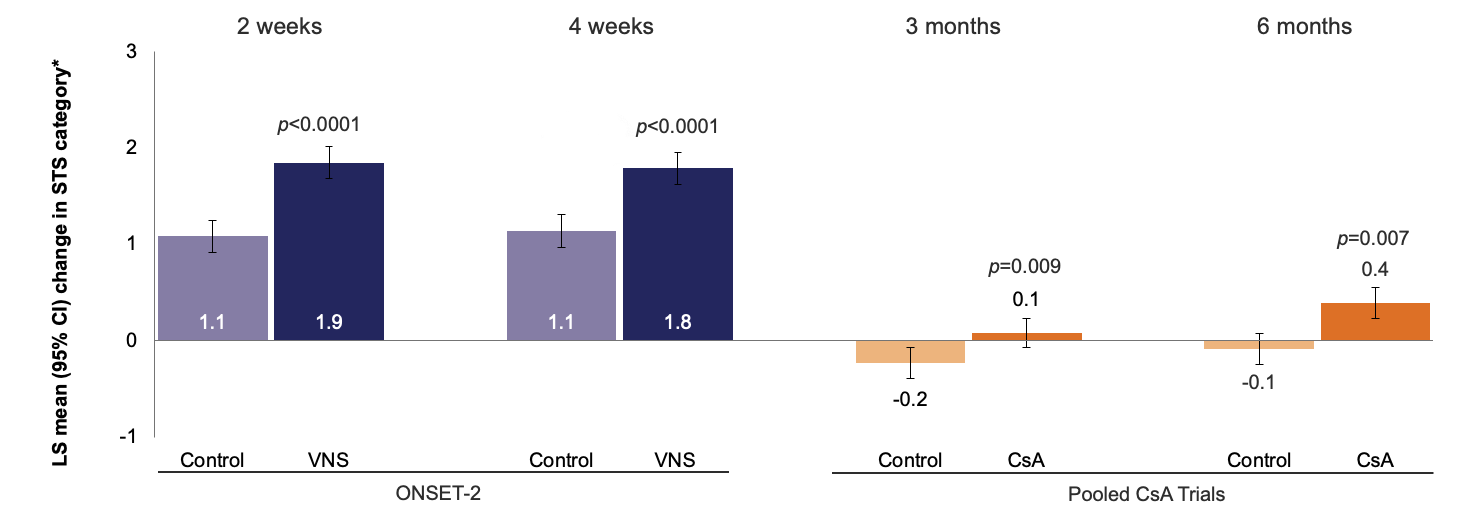
Weighted results of LS mean change from baseline in STS with anesthesia for ONSET-2 and CsA trials. p-values are for treatment vs control at each time point; *In the CsA trials, the continuous variable STS was reduced to a categorical 5-point variable as follows: 1 (<3 mm), 2 (3–6 mm), 3 (7–10 mm), 4 (11–14 mm), and 5 (>14 mm). All ONSET-2 data were recoded to match this scale.
CI, confidence interval; CsA, cyclosporin A; LS, least squares; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
CI, confidence interval; CsA, cyclosporin A; LS, least squares; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
ONSET-2 vs CsA MAIC Outcomes
Efficacy Outcome Measures – Common Endpoint
Mean change in STS (with anesthesia) category* from baseline
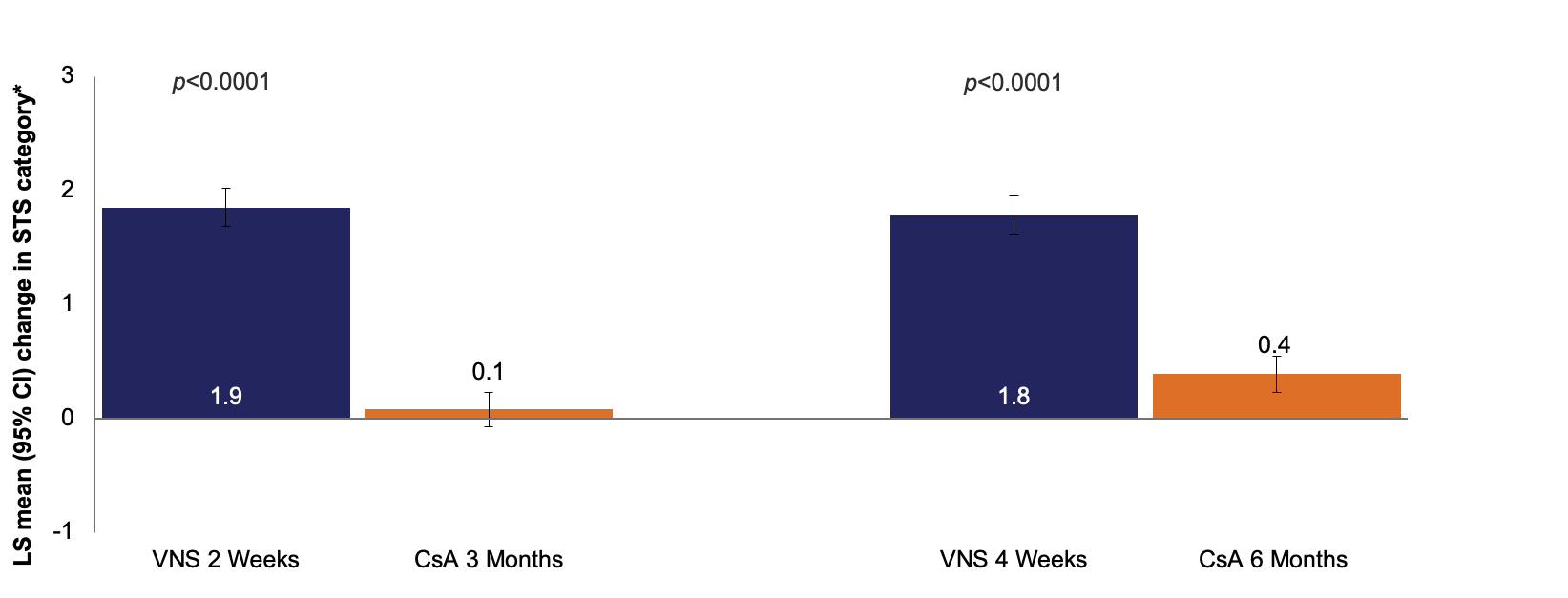
Results of ANOVA models assessing the LS mean change from baseline in STS with anesthesia for ONSET-2 and CsA trials. p-values are for treatment vs control at each time point;*In the CsA trials, the continuous variable STS was reduced to a categorical 5-point variable as follows: 1 (<3 mm), 2 (3–6 mm), 3 (7–10 mm), 4 (11–14 mm), and 5 (>14 mm). CsA data was pooled from -002 and -003 CsA phase 3 studies. All ONSET-2 data were recoded to match this scale.
ANOVA, analysis of variance; CI, confidence interval; CsA, cyclosporin A; LS, least squares; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
ANOVA, analysis of variance; CI, confidence interval; CsA, cyclosporin A; LS, least squares; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
ONSET-2 vs CsA Trials Outcomes
Efficacy Outcome Measures – Common Endpoint
Proportion of subjects with ≥10 mm improvement in STS with anesthesia from baseline
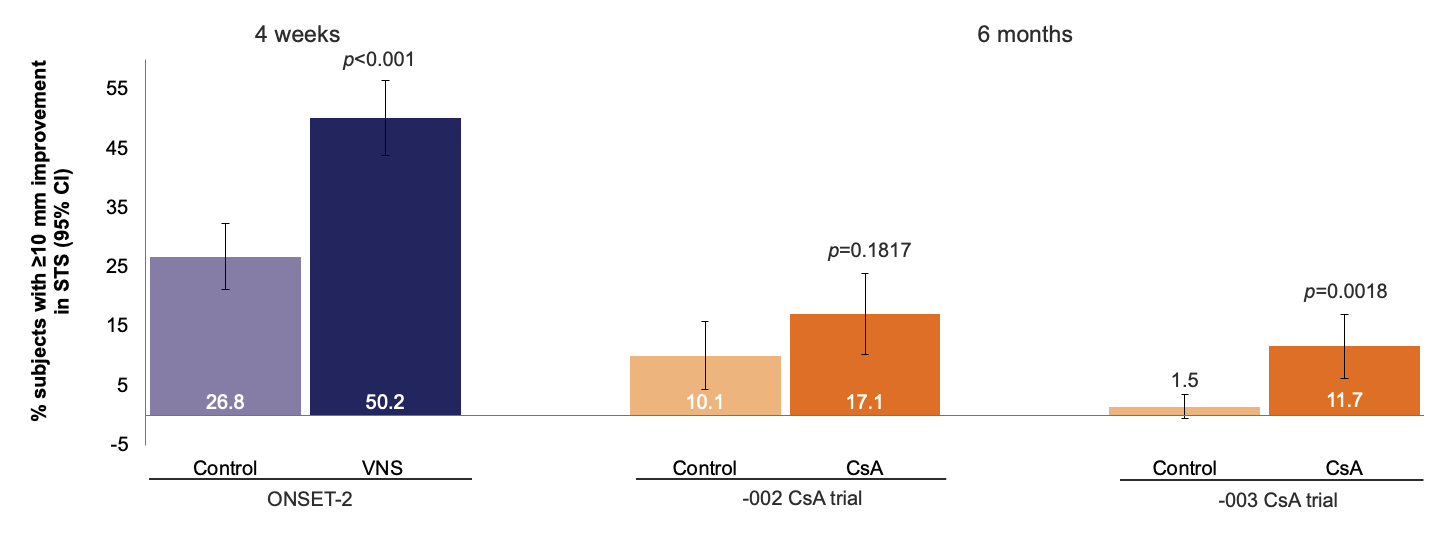
Weighted results of responder analyses of the proportions of subjects with a ≥10 mm improvement in STS with anesthesia for ONSET-2 and CsA trials. p-values are for treatment vs control at each time point.
CI, confidence interval; CsA, cyclosporin A; LS, least squares; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
CI, confidence interval; CsA, cyclosporin A; LS, least squares; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
ONSET-2 vs CsA MAIC Outcomes
Efficacy Outcome Measures – Common Endpoint
Proportion of subjects with ≥10 mm improvement in STS with anesthesia from baseline
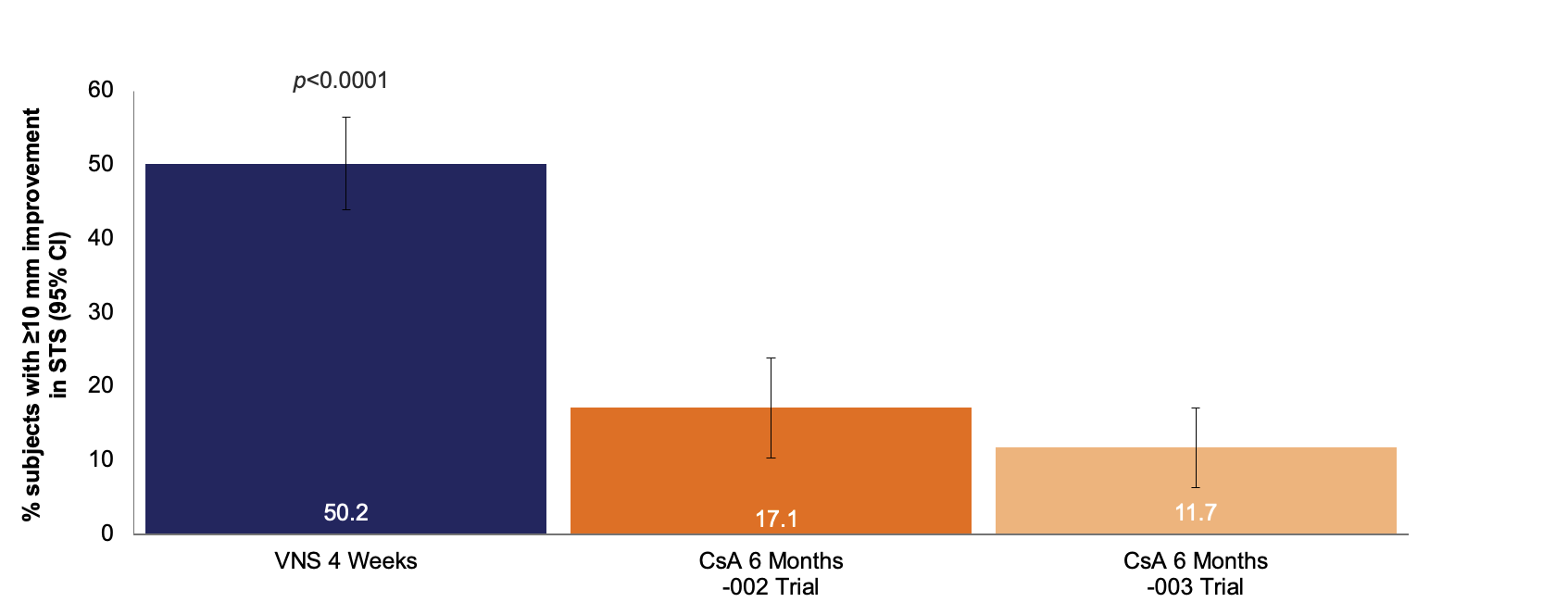
Results of responder analyses of the proportions of subjects with a ≥10 mm improvement in STS with anesthesia for ONSET-2 and CsA trials. p-values are for treatment vs control at each time point.
ANOVA, analysis of variance; CI, confidence interval; CsA, cyclosporin A; LS, least squares; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.
ANOVA, analysis of variance; CI, confidence interval; CsA, cyclosporin A; LS, least squares; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
Visco DM. et al. J Manag Care Spec Pharm. 2022;28:892–902.

