Multicenter, Randomized, Controlled, Double-Masked, Clinical Trial to Evaluate the Efficacy and Safety of OC-01 (Varenicline) Nasal Spray on Signs and Symptoms of Dry Eye Disease
| Molecule | Varenicline solution nasal spray (formerly known as OC-01) |
| Therapeutic focus | Dry eye disease |
| Phase | 3 |
| Status | Completed |
| Participants | 758 |
| Completion Date | February 5, 2021 |
https://clinicaltrials.gov/study/NCT04036292.

Trial Design1
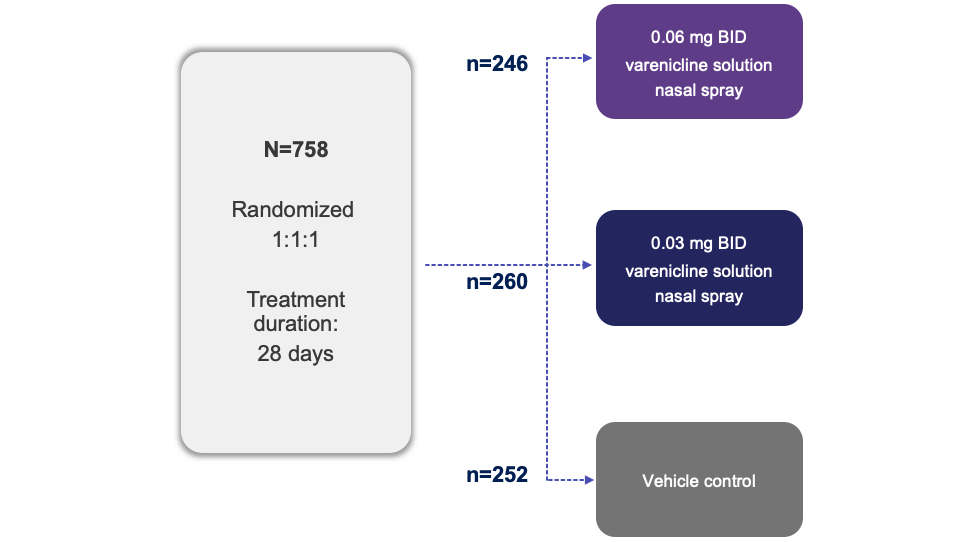
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2
BID, twice daily; FDA, Food and Drug Administration.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
BID, twice daily; FDA, Food and Drug Administration.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Baseline Characteristics1
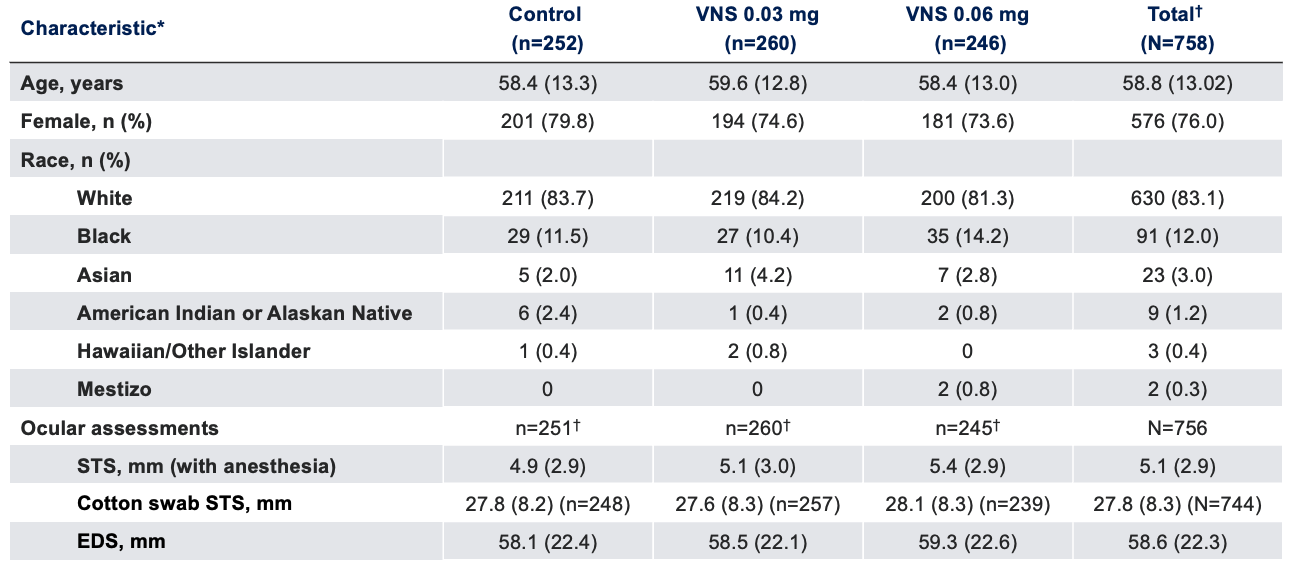
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 *Data presented as mean (SD), unless otherwise specified;†Oyster Point Pharma, Inc. Data on file.
EDS, eye dryness score; FDA, Food and Drug Administration; SD, standard deviation; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
EDS, eye dryness score; FDA, Food and Drug Administration; SD, standard deviation; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Primary Endpoint1
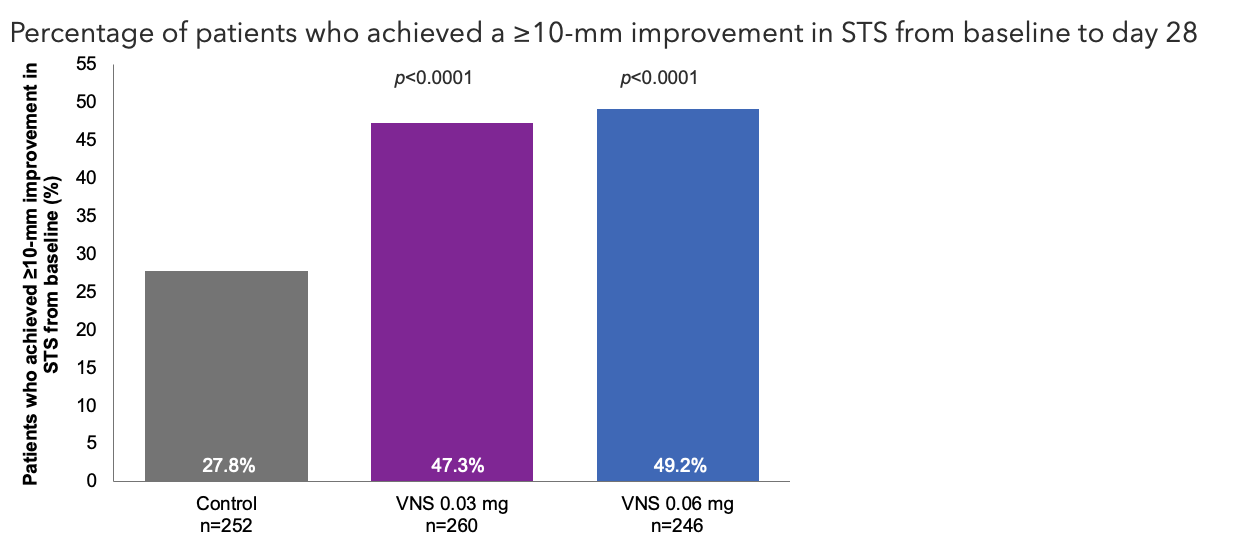
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 ITT population. Cochran-Mantel-Haenszel test controlling for study site, baseline STS, and baseline EDS. All comparisons made with the control group.1
EDS, eye dryness score; FDA, Food and Drug Administration; ITT, intention-to-treat; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
EDS, eye dryness score; FDA, Food and Drug Administration; ITT, intention-to-treat; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Secondary Endpoint1
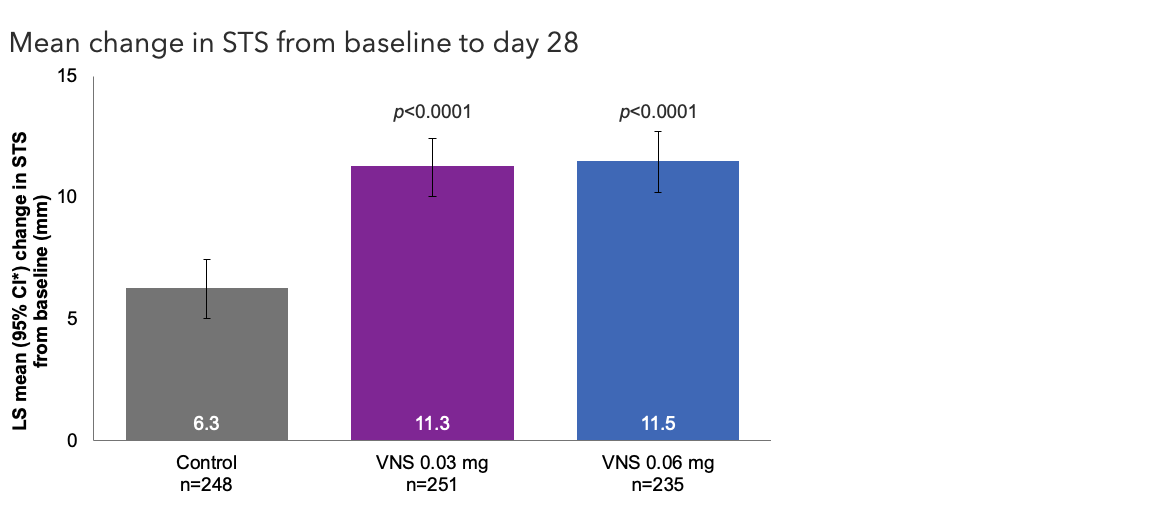
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 ITT population. Pairwise treatment group differences were compared using ANCOVA models with treatment group, study site, baseline STS, and baseline EDS as covariates. All comparisons made with the control group. Nominalp-values.1 *Oyster Point Pharma, Inc. Data on file.
ANCOVA, analysis of covariance; CI, confidence interval; EDS, eye dryness score; FDA, Food and Drug Administration; ITT, intention-to-treat; LS, least squares; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
ANCOVA, analysis of covariance; CI, confidence interval; EDS, eye dryness score; FDA, Food and Drug Administration; ITT, intention-to-treat; LS, least squares; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Secondary Endpoint1
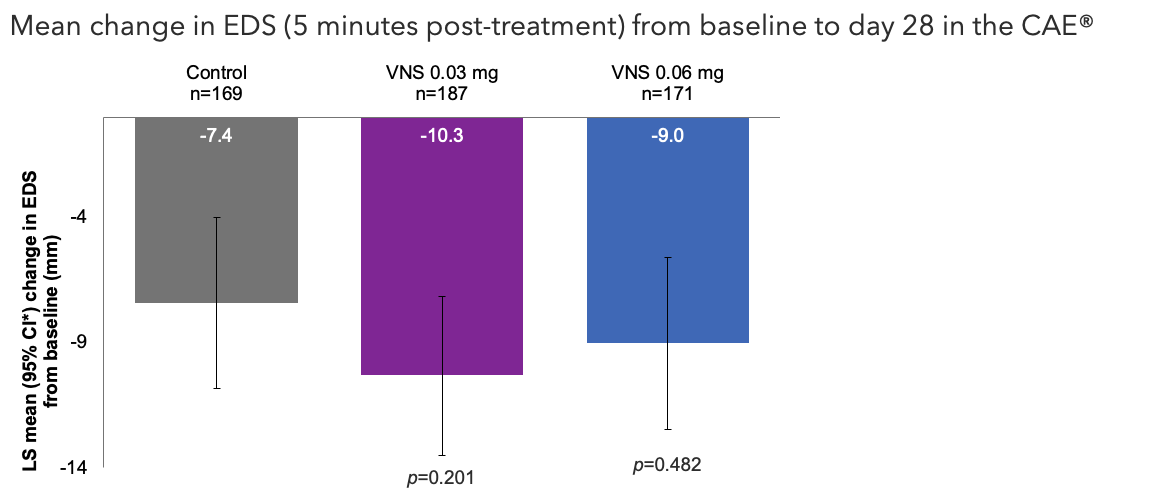
Note: The FDA-approved dosage of Varenicline solution nasal spray is 0.03 mg BID.2 Modified ITT population (excluded 2 sites because of their inability to collect specific study data due to the coronavirus disease 2019 pandemic). Pairwise treatment group differences were compared using ANCOVA models with treatment group, study site, baseline STS, and baseline EDS as covariates. All comparisons made with the control group. Nominal p-values.1 Oyster Point Pharma, Inc. Data on file.
ANCOVA, analysis of covariance; CAE®, controlled adverse environment; CI, confidence interval; EDS, eye dryness score; FDA, Food and Drug Administration; ITT, intention-to-treat; LS, least squares; STS, Schirmer's test score; VNS, Varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(Varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
ANCOVA, analysis of covariance; CAE®, controlled adverse environment; CI, confidence interval; EDS, eye dryness score; FDA, Food and Drug Administration; ITT, intention-to-treat; LS, least squares; STS, Schirmer's test score; VNS, Varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(Varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Secondary Endpoint1
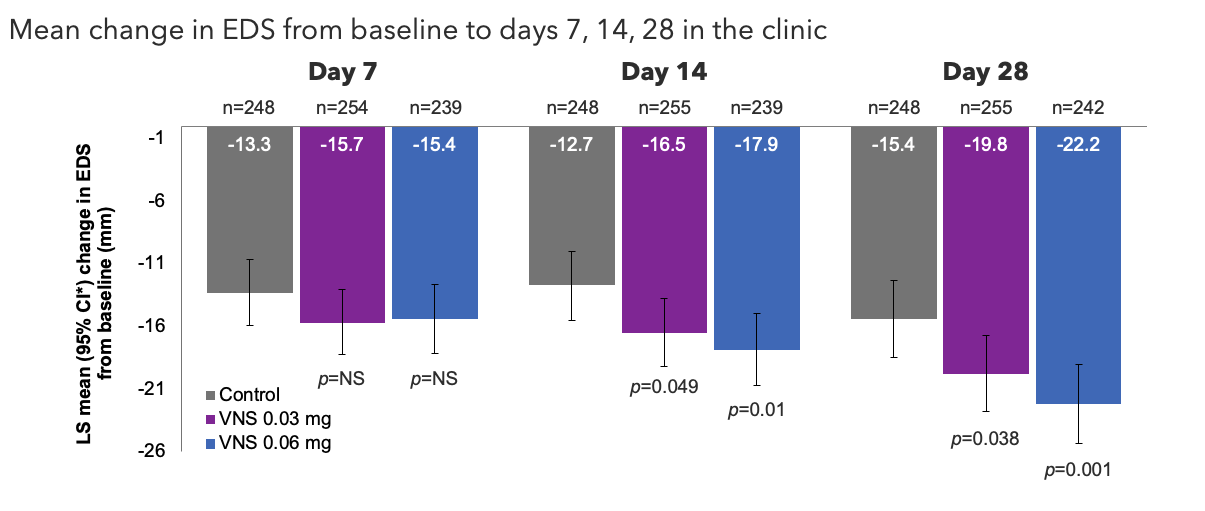
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 ITT population. Pairwise treatment group differences were compared using ANCOVA models with treatment group, study site, baseline STS, and baseline EDS as covariates. All comparisons made with the control group. Nominalp-values due to closed hierarchical testing procedure.1*Oyster Point Pharma, Inc. Data on file.
ANCOVA, analysis of covariance; CI, confidence interval; EDS, eye dryness score; FDA, Food and Drug Administration; ITT, intention-to-treat; LS, least squares; NS, not significant; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
ANCOVA, analysis of covariance; CI, confidence interval; EDS, eye dryness score; FDA, Food and Drug Administration; ITT, intention-to-treat; LS, least squares; NS, not significant; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Safety Outcome Measures1
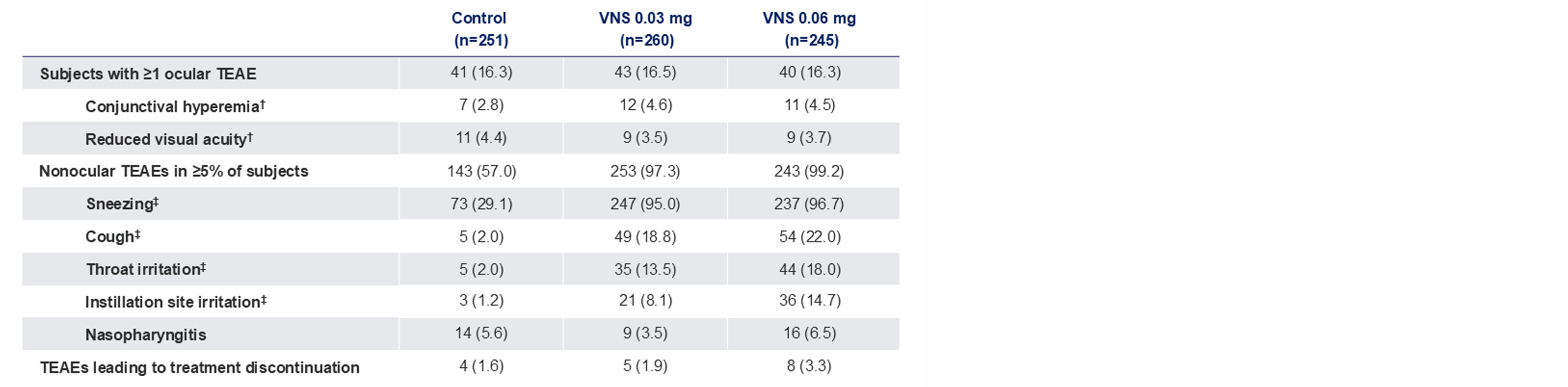
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 *Data presented as n (%). No clinically significant findings for intranasal examination, slitlamp biomicroscopy, or BCVA assessment. 3 deaths occurred during the study (during the post-treatment follow-up period) as a result of adverse events not considered related to the study drug (pneumonia, cerebrovascular accident, coronavirus infection). No patients reported serious TEAEs considered to be related to the study drug; †The most commonly reported ocular TEAEs. Most of these events were not considered related to the study drug; ‡The most commonly reported nonocular TEAEs. Most events were considered mild. The incidences of all other nonocular TEAEs were similar across treatment groups.
BCVA, best-corrected visual acuity; FDA, Food and Drug Administration; TEAE, treatment-emergent adverse event; VNS, varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
BCVA, best-corrected visual acuity; FDA, Food and Drug Administration; TEAE, treatment-emergent adverse event; VNS, varenicline solution nasal spray.
1. Wirta D. et al.Ophthalmology. 2022;129:379–387; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Safety Outcome Measures
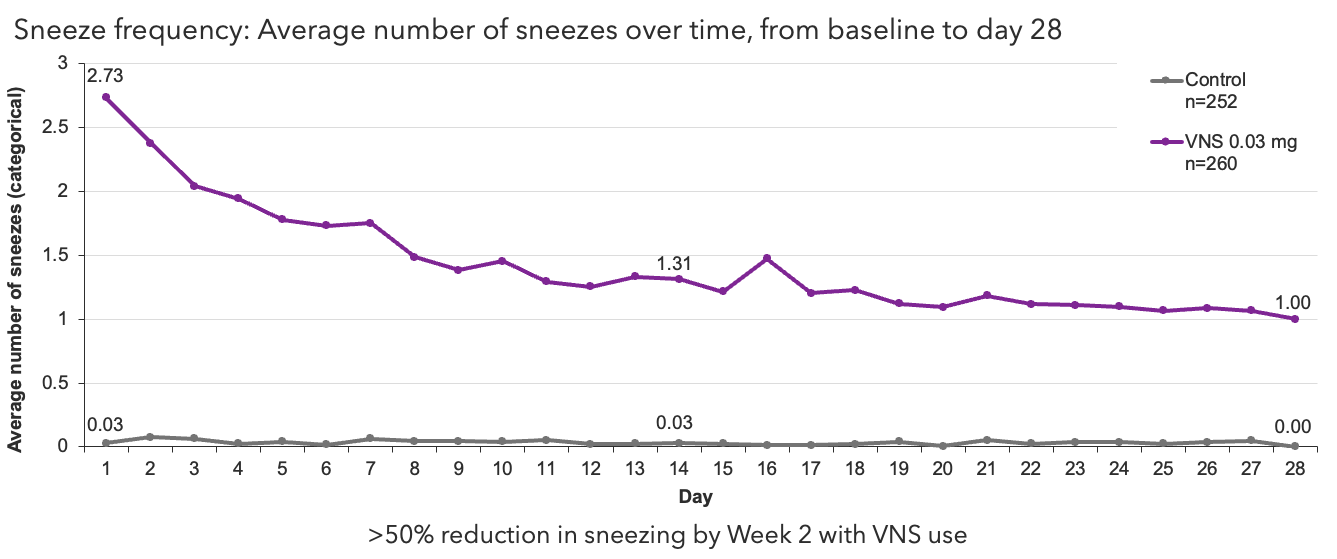
Categories for number of sneezes after study drug delivery: 0 (no sneezes), 1.5 (1–2 sneezes), 4 (3–5 sneezes), and 7 (≥6 sneezes).
Oyster Point Pharma, Inc. Data on file.
VNS, varenicline solution nasal spray.
Oyster Point Pharma, Inc. Data on file.
VNS, varenicline solution nasal spray.

