Multicenter, Randomized, Controlled, Double-Masked Clinical Trial to Evaluate the Efficacy of OC-01 Nasal Spray on Signs and Symptoms of Dry Eye Disease
| Molecule | Varenicline solution nasal spray (formerly known as OC-01) |
| Therapeutic focus | Dry eye disease |
| Phase | 2b |
| Status | Completed |
| Participants | 182 |
| Completion Date | September 26, 2018 |
https://clinicaltrials.gov/study/NCT03636061.

Trial Design1
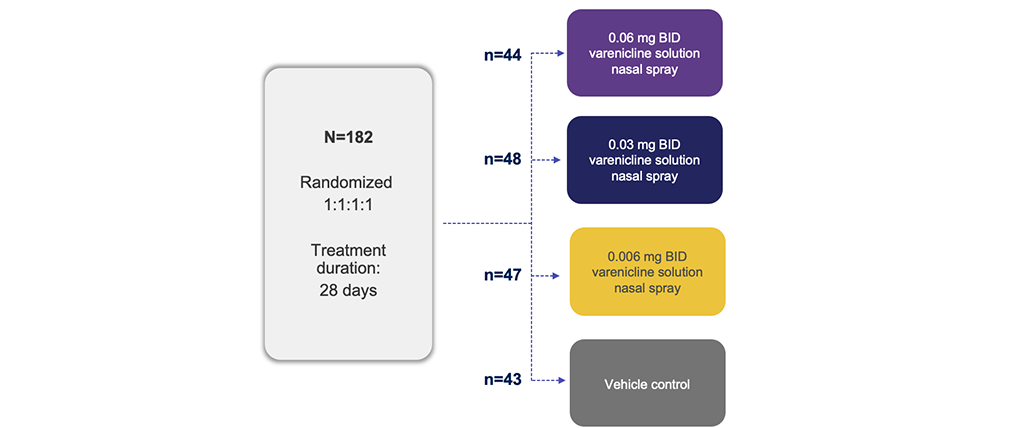
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2
BID, twice daily; FDA, Food and Drug Administration.
1. Wirta D. et al.Cornea. 2022;41:1207–1216; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
BID, twice daily; FDA, Food and Drug Administration.
1. Wirta D. et al.Cornea. 2022;41:1207–1216; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Baseline Characteristics1
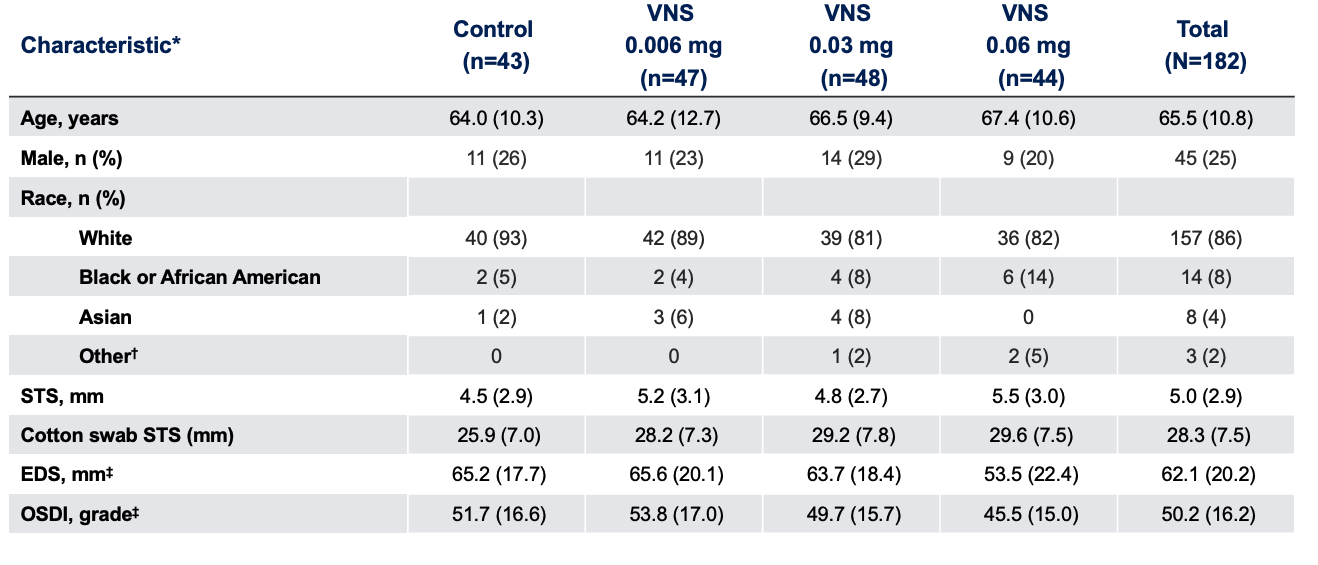
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 *Data presented as mean (SD), unless otherwise specified;†Includes American Indian or Alaska Native, and Native Hawaiian or other Pacific Islander;‡Assessment relates to both eyes.
EDS, eye dryness score; FDA, Food and Drug Administration; OSDI, Ocular Surface Disease Index; SD, standard deviation; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
1. Wirta D. et al. Cornea. 2022;41:1207–1216; 2. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
EDS, eye dryness score; FDA, Food and Drug Administration; OSDI, Ocular Surface Disease Index; SD, standard deviation; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
1. Wirta D. et al. Cornea. 2022;41:1207–1216; 2. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Primary Endpoint1
Mean change in STS in the study eye from baseline to day 28
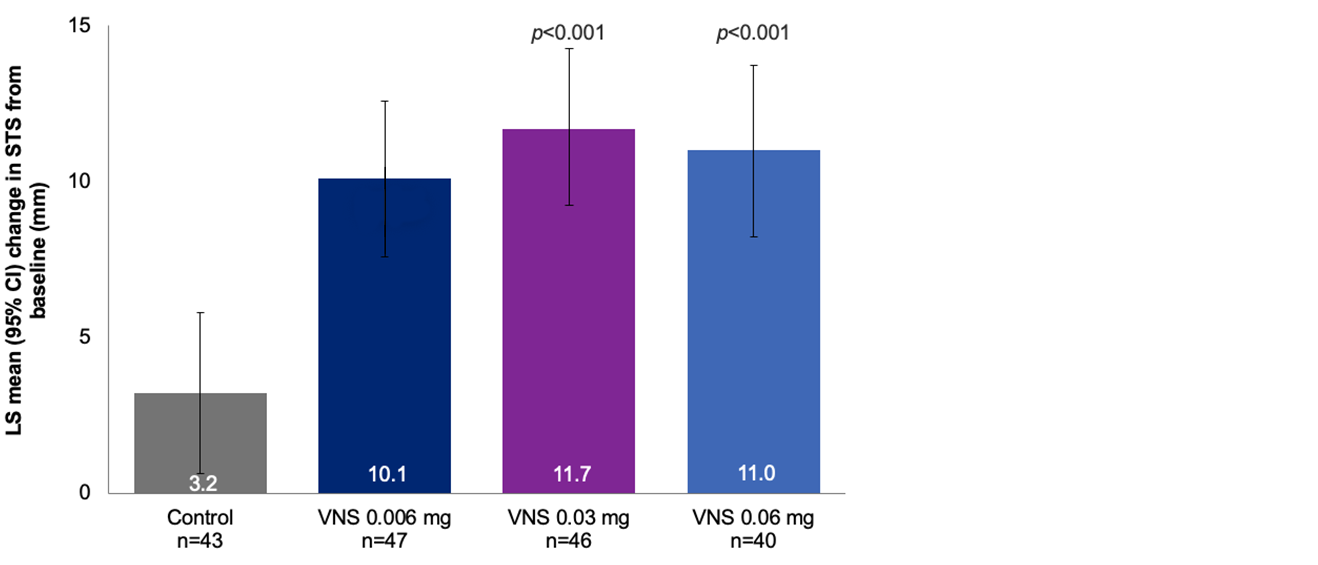
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 LS means were derived from ANCOVA model with treatment, site, baseline STS, and STS with a cotton swab stimulation as covariates.3 The results are derived from a posthoc analysis. LOCF imputation method is used.1,3 All comparisons made with the control group. No formal statistical comparisons were performed for the 0.006 mg dose versus control.3
ANCOVA, analysis of covariance; CI, confidence interval; FDA, Food and Drug Administration; LOCF; last observation carried forward; LS, least squares; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
1. ONSET-1 Oyster Point Pharma, Inc. Data on file; 2. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf; 3. Wirta D. et al. Cornea. 2022;41:1207–1216.
ANCOVA, analysis of covariance; CI, confidence interval; FDA, Food and Drug Administration; LOCF; last observation carried forward; LS, least squares; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
1. ONSET-1 Oyster Point Pharma, Inc. Data on file; 2. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf; 3. Wirta D. et al. Cornea. 2022;41:1207–1216.
Efficacy Outcome Measures – Exploratory Endpoint1
Percentage of subjects with ≥10 mm change from baseline in STS at day 28
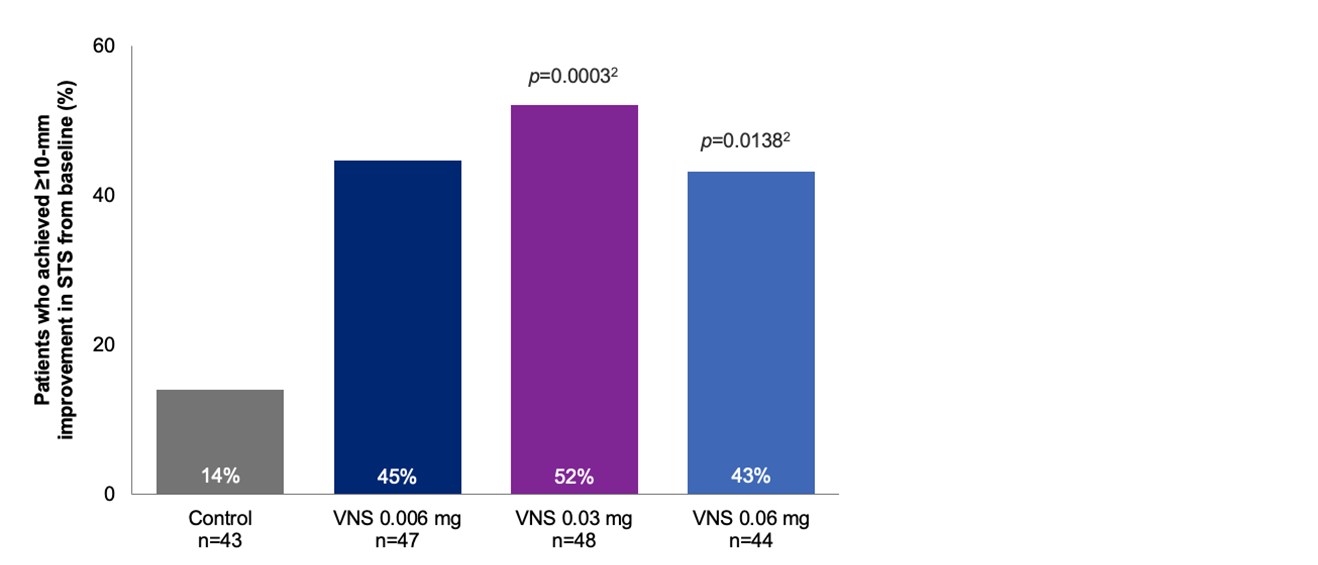
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.3 The results are derived from a posthoc analysis. All comparisons made with the control group. No formal statistical comparisons were performed for the 0.006 mg dose versus control.2
FDA, Food and Drug Administration; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
1. ONSET-1 Oyster Point Pharma, Inc. Data on file; 2. Wirta D. et al. Cornea. 2022;41:1207–1216; 3. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
FDA, Food and Drug Administration; STS, Schirmer’s test score; VNS, varenicline solution nasal spray.
1. ONSET-1 Oyster Point Pharma, Inc. Data on file; 2. Wirta D. et al. Cornea. 2022;41:1207–1216; 3. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Secondary Endpoint1
Mean change in EDS from baseline in clinic to day 281,2
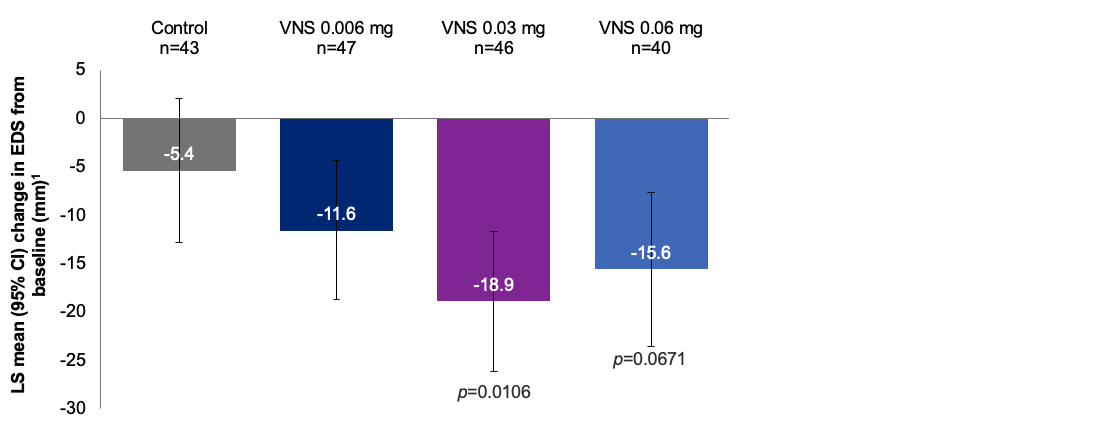
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.3 LS means were derived from ANCOVA model with treatment, site, and baseline EDS as covariates. The results are derived from a posthoc analysis. All comparisons made with the control group. No formal statistical comparisons were performed for the 0.006 mg dose versus control.2
ANCOVA, analysis of covariance; CI, confidence interval; EDS, eye dryness score; LS, least squares; VNS, varenicline solution nasal spray.
1. ONSET-1 Oyster Point Pharma, Inc. Data on file; 2. Wirta D. et al. Cornea. 2022;41:1207–1216; 3. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
ANCOVA, analysis of covariance; CI, confidence interval; EDS, eye dryness score; LS, least squares; VNS, varenicline solution nasal spray.
1. ONSET-1 Oyster Point Pharma, Inc. Data on file; 2. Wirta D. et al. Cornea. 2022;41:1207–1216; 3. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Secondary Endpoint1
Mean change in EDS from baseline to day 21 (5 minutes post-treatment) in the CAE®
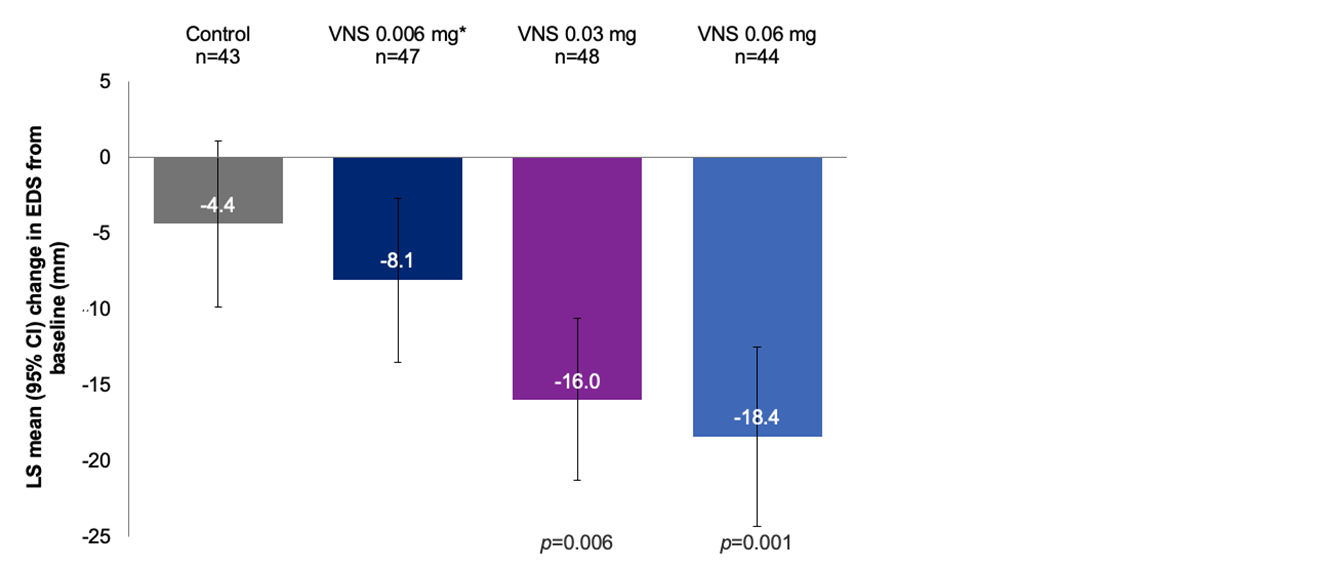
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 Least squares means were derived from ANCOVA model with treatment, site, and baseline EDS as covariates. The results are derived from a post hoc analysis. All comparisons made with the control group. No formal statistical comparisons were performed for the 0.006 mg dose versus control.1 *Oyster Point Pharma, Inc. Data on file.
ANCOVA, analysis of covariance; CAE®, controlled adverse environment; CI, confidence interval; EDS, eye dryness score; VNS, varenicline solution nasal spray.
1. Wirta D. et al. Cornea. 2022;41:1207–1216; 2. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
ANCOVA, analysis of covariance; CAE®, controlled adverse environment; CI, confidence interval; EDS, eye dryness score; VNS, varenicline solution nasal spray.
1. Wirta D. et al. Cornea. 2022;41:1207–1216; 2. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Safety Outcome Measures*1
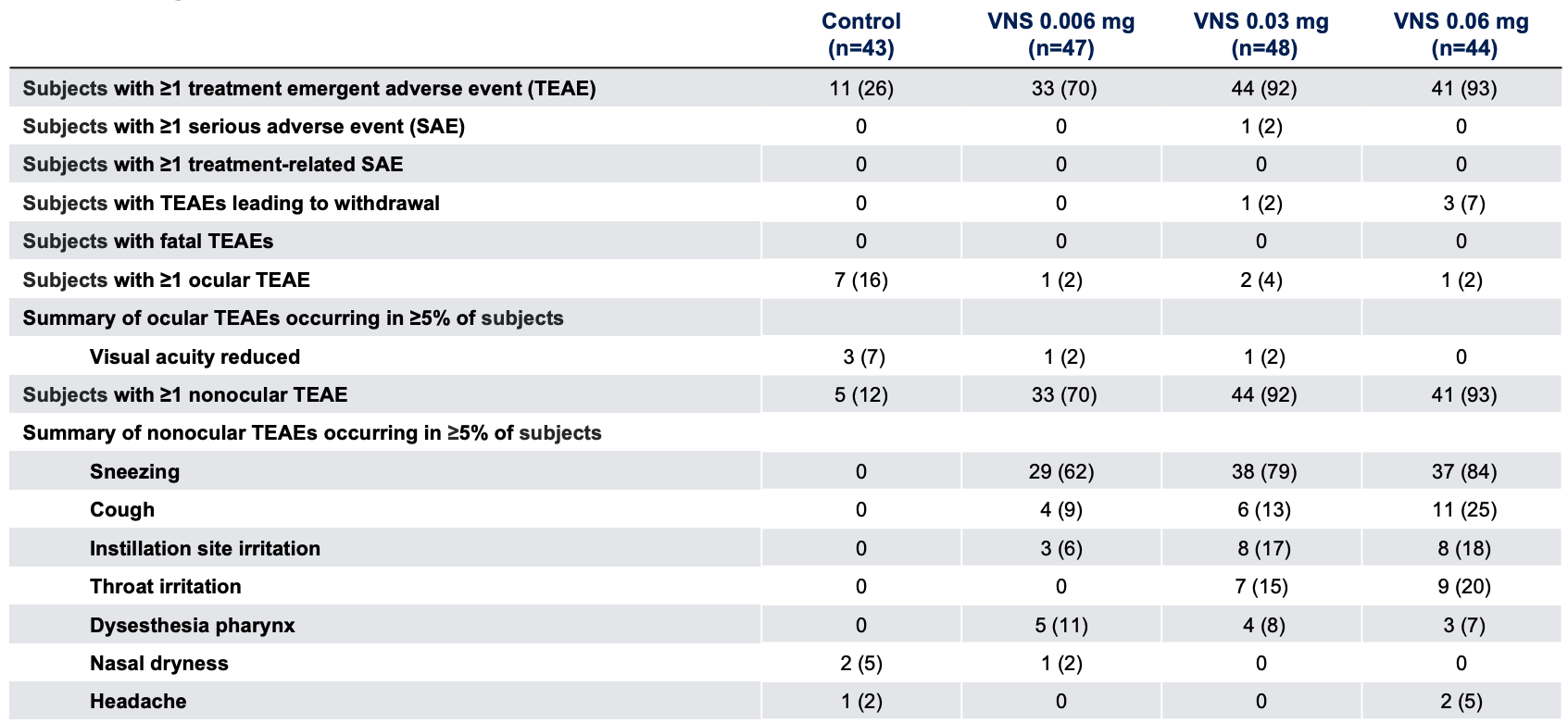
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 *Data presented as n (%). VNS, varenicline solution nasal spray.
1. Wirta D. et al. Cornea. 2022;41:1207–1216; 2. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
1. Wirta D. et al. Cornea. 2022;41:1207–1216; 2. TYRVAYA® (varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.

