Randomized, Controlled, Single-Masked Clinical Trial to Evaluate the Chronic Efficacy of OC-01 Nasal Spray on Signs of Dry Eye Disease
| Molecule | Varenicline solution nasal spray (formerly known as OC-01) |
| Therapeutic focus | Dry eye disease |
| Phase | 2 |
| Status | Completed |
| Participants | 123 |
| Completion Date | January 7, 2020 |
Note: Single-site study. All subjects were Hispanic or Latino.
https://clinicaltrials.gov/study/NCT03873246.

Trial Design1
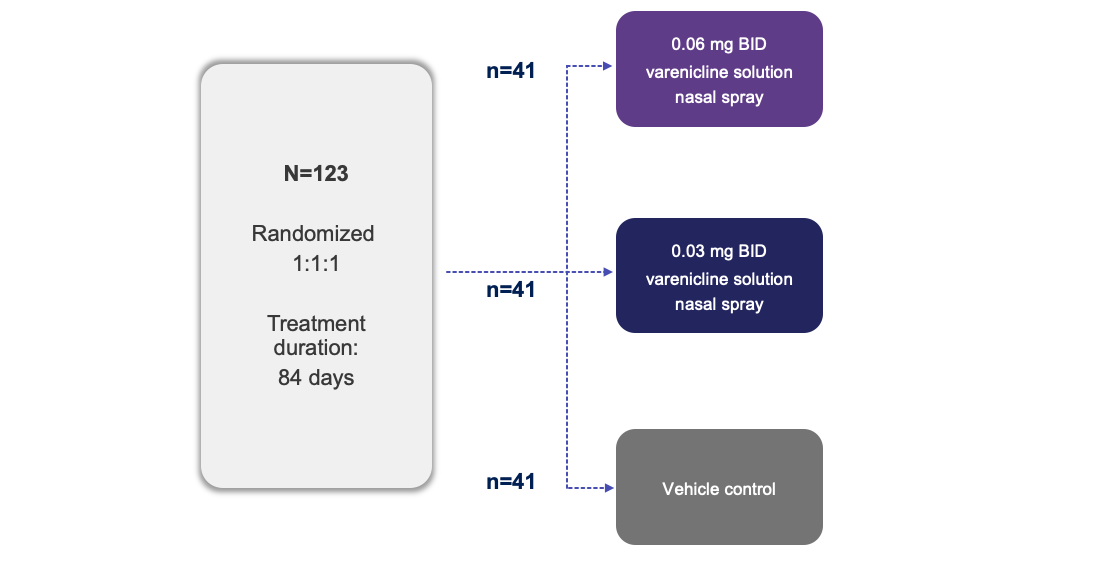
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2
BID, twice daily; FDA, Food and Drug Administration.
1. Quiroz-Mercado H. et al.Ocul Surf. 2022;24:15–21; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
BID, twice daily; FDA, Food and Drug Administration.
1. Quiroz-Mercado H. et al.Ocul Surf. 2022;24:15–21; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Baseline Characteristics1
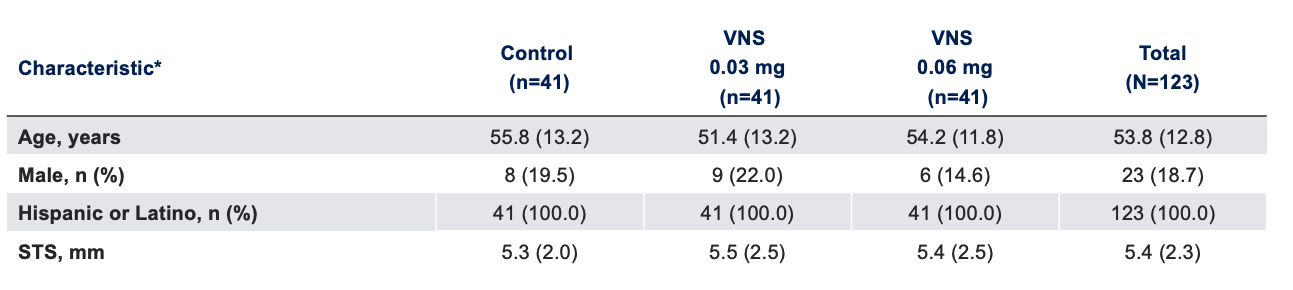
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 *Data presented as mean (SD), unless otherwise specified.
FDA, Food and Drug Administration; SD, standard deviation; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Quiroz-Mercado H. et al.Ocul Surf. 2022;24:15–21; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
FDA, Food and Drug Administration; SD, standard deviation; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Quiroz-Mercado H. et al.Ocul Surf. 2022;24:15–21; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Primary Endpoint1
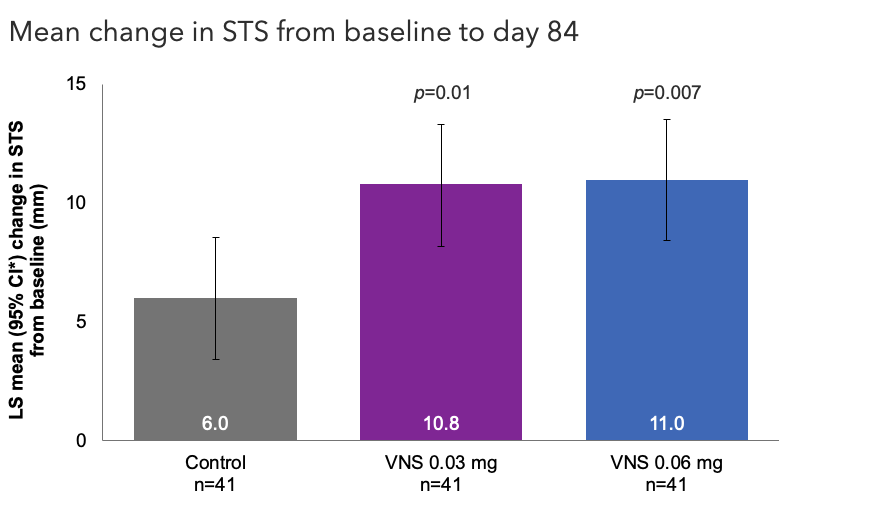
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 Analyzed using an ANCOVA model with baseline STS and treatment group as covariates. All comparisons made with the control group.1 *Oyster Point Pharma, Inc. Data on file.
ANCOVA, analysis of covariance; CI, confidence interval; FDA, Food and Drug Administration; LS, least squares; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Quiroz-Mercado H. et al.Ocul Surf. 2022;24:15–21; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
ANCOVA, analysis of covariance; CI, confidence interval; FDA, Food and Drug Administration; LS, least squares; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Quiroz-Mercado H. et al.Ocul Surf. 2022;24:15–21; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Secondary Endpoint1
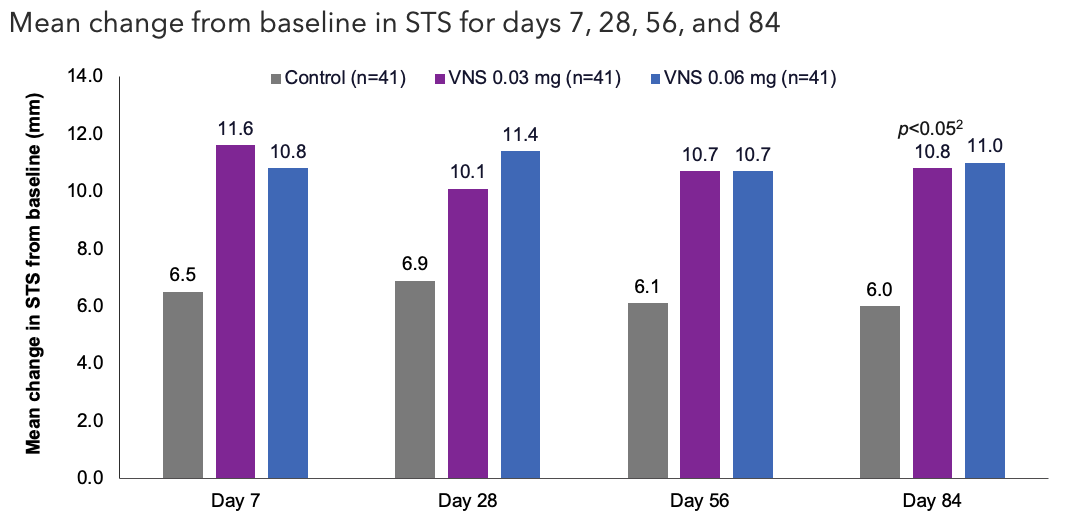
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.3 Analyzed using an ANCOVA model with baseline STS and treatment group as covariates. All comparisons made with the control group.2 VNS 0.03 mg group mean baseline STS 5.5 mm (n=41); VNS 0.06 mg group mean baseline STS 5.4 mm (n=41); control group mean baseline STS 5.3 mm (n=41).1 All randomized and treated patients were included in the analysis and missing data were imputed using last-available data.1,2
ANCOVA, analysis of covariance; FDA, Food and Drug Administration; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. MYSTIC Oyster Point Pharma, Inc. Data on file; 2. Quiroz-Mercado H. et al.Ocul Surf. 2022;24:15–21; 3. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
ANCOVA, analysis of covariance; FDA, Food and Drug Administration; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. MYSTIC Oyster Point Pharma, Inc. Data on file; 2. Quiroz-Mercado H. et al.Ocul Surf. 2022;24:15–21; 3. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Safety Outcome Measures*1
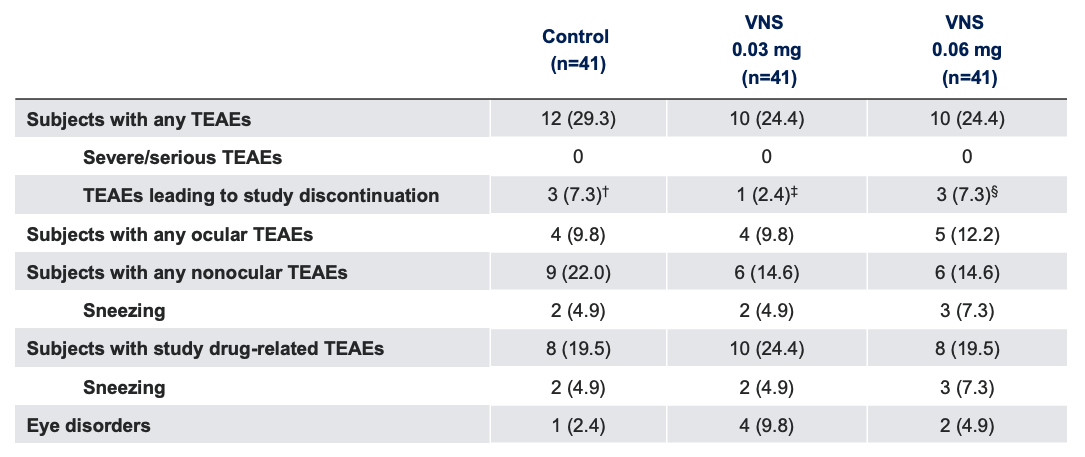
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.3 *Data presented as n (%). TEAEs >5% are reported. No clinically relevant changes from baseline in BCVA, slitlamp biomicroscopy findings, pupil diameter, or intranasal examinations were observed. No deaths were reported; †Depression on day 30. Nausea, dizziness, and nasal discomfort on day 1. Chest pain and dyspnea on day 1; ‡Headache on day 1; §Nausea on day 1. Headache on day 13; eye irritation and headache on day 1.
BCVA, best-corrected visual acuity; FDA, Food and Drug Administration; TEAE, treatment-emergent adverse event; VNS, varenicline solution nasal spray.
1. Quiroz-Mercado H. et al.Ocul Surf. 2022;24:15–21; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
BCVA, best-corrected visual acuity; FDA, Food and Drug Administration; TEAE, treatment-emergent adverse event; VNS, varenicline solution nasal spray.
1. Quiroz-Mercado H. et al.Ocul Surf. 2022;24:15–21; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.

