A Single-Center, Randomized, Controlled, Masked Clinical Trial to Evaluate the Efficacy of OC-01 Nasal Spray on Goblet Cell and Meibomian Gland Stimulation
| Molecule | Varenicline solution nasal spray (formerly known as OC-01) |
| Therapeutic focus | Dry eye disease |
| Phase | 2 |
| Status | Completed |
| Participants | 18 |
| Completion Date | September 30, 2019 |
Note: This clinical trial was not included in the submission which resulted in Tyrvaya® approval.
https://clinicaltrials.gov/study/NCT03688802.

Trial Design1
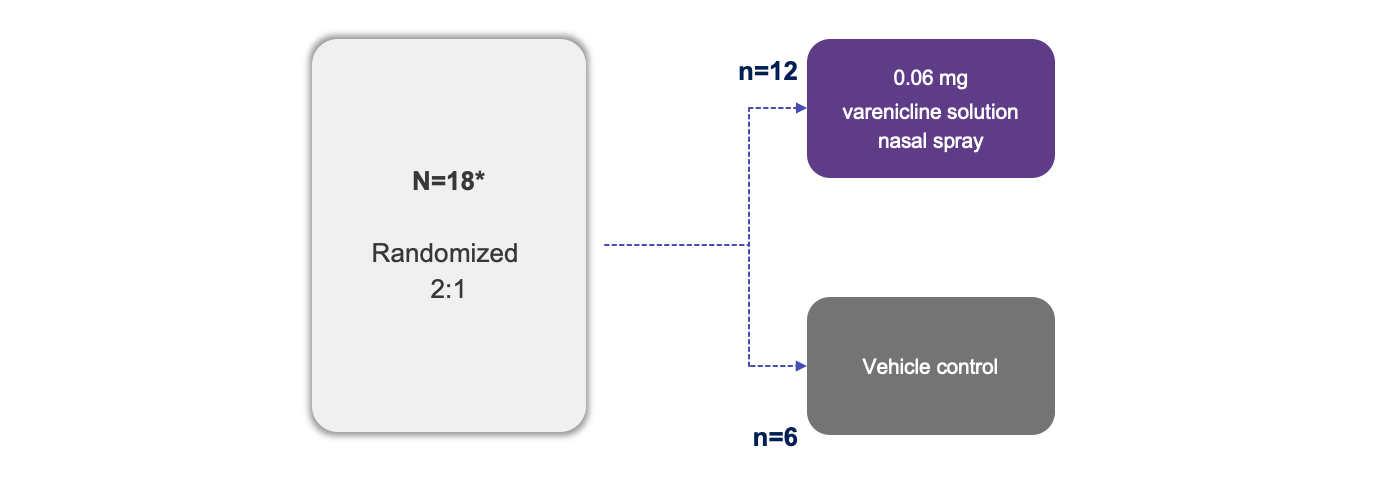
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 This clinical trial was not included in the submission which resulted in Tyrvaya® approval.
*Target enrollment was 45. However, enrollment was slower than expected, and the study was ended prior to completing target enrollment.
FDA, Food and Drug Administration.
1. Dieckmann G.M. et al.Ophthalmol Ther. 2022;11:1551–1561; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
*Target enrollment was 45. However, enrollment was slower than expected, and the study was ended prior to completing target enrollment.
FDA, Food and Drug Administration.
1. Dieckmann G.M. et al.Ophthalmol Ther. 2022;11:1551–1561; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Baseline Characteristics1
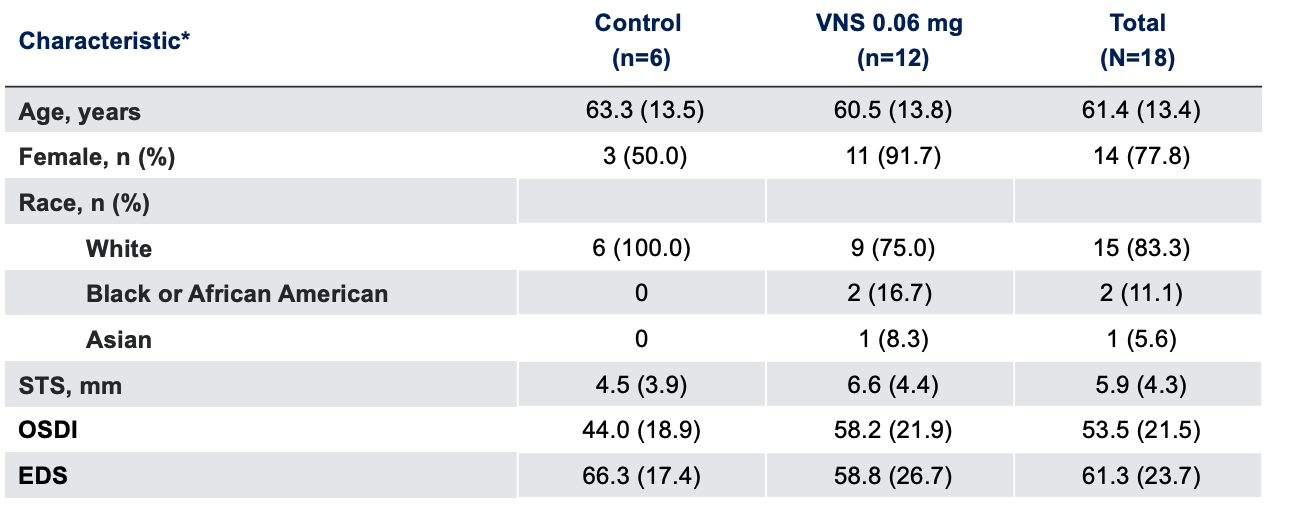
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 *Data presented as mean (SD), unless otherwise specified.
EDS, eye dryness score; FDA, Food and Drug Administration; OSDI, Ocular Surface Disease Index; SD, standard deviation; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Dieckmann G.M. et al.Ophthalmol Ther. 2022;11:1551–1561; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
EDS, eye dryness score; FDA, Food and Drug Administration; OSDI, Ocular Surface Disease Index; SD, standard deviation; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
1. Dieckmann G.M. et al.Ophthalmol Ther. 2022;11:1551–1561; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Primary Endpoint1
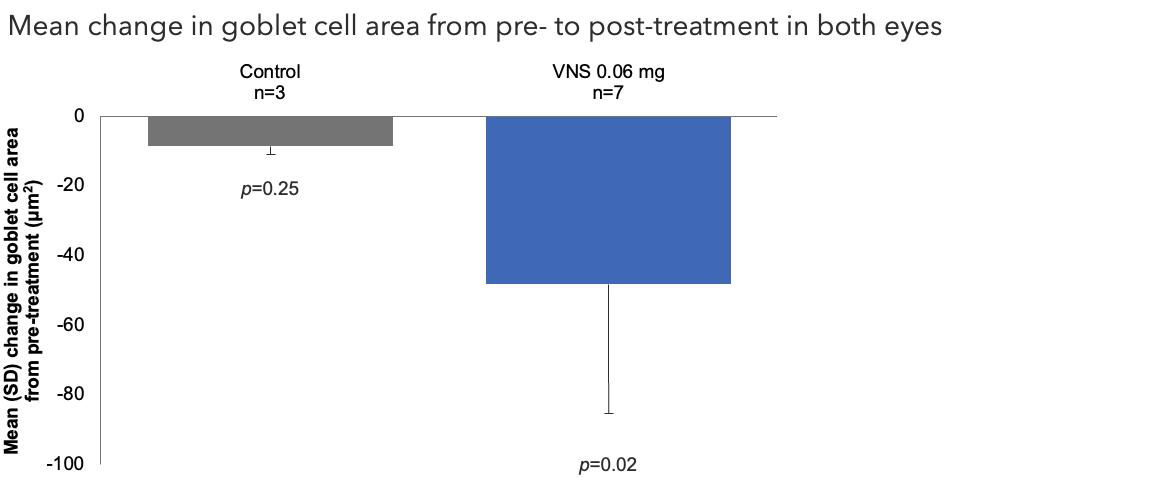
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 Outcomes were compared using an ANCOVA model with treatment group and baseline measurements (meibomian cell area, goblet cell area, and goblet cell perimeter) as covariates.1
ANCOVA, analysis of covariance; FDA, Food and Drug Administration; SD, standard deviation; VNS, varenicline solution nasal spray.
1. Dieckmann G.M. et al.Ophthalmol Ther. 2022;11:1551–1561; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
ANCOVA, analysis of covariance; FDA, Food and Drug Administration; SD, standard deviation; VNS, varenicline solution nasal spray.
1. Dieckmann G.M. et al.Ophthalmol Ther. 2022;11:1551–1561; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Efficacy Outcome Measures – Primary Endpoint1
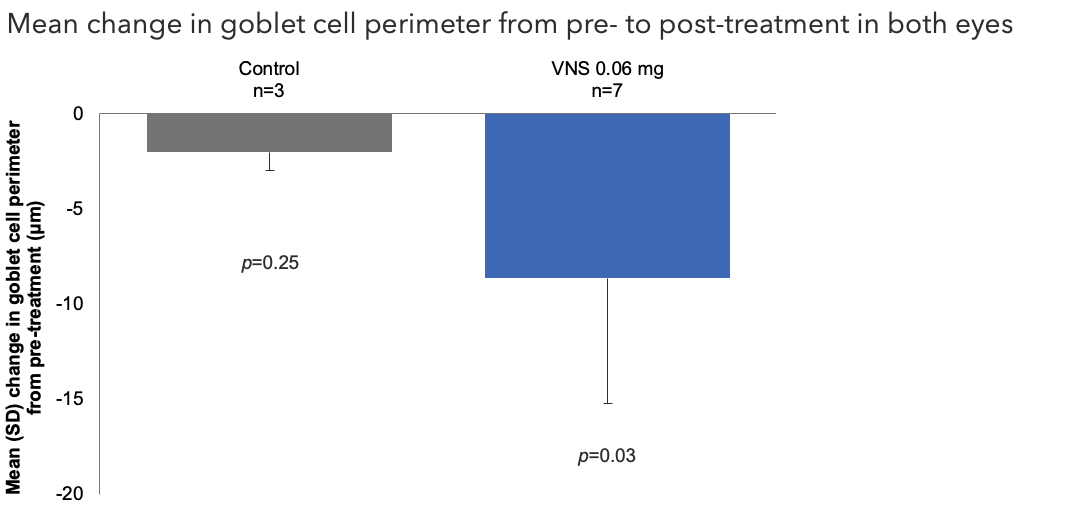
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 Outcomes were compared using an ANCOVA model with treatment group and baseline measurements (meibomian cell area, goblet cell area, and goblet cell perimeter) as covariates.1
ANCOVA, analysis of covariance; FDA, Food and Drug Administration; SD, standard deviation; VNS, varenicline solution nasal spray.
1. Dieckmann G.M. et al.Ophthalmol Ther. 2022;11:1551–1561; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
ANCOVA, analysis of covariance; FDA, Food and Drug Administration; SD, standard deviation; VNS, varenicline solution nasal spray.
1. Dieckmann G.M. et al.Ophthalmol Ther. 2022;11:1551–1561; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
Safety Outcome Measures*1
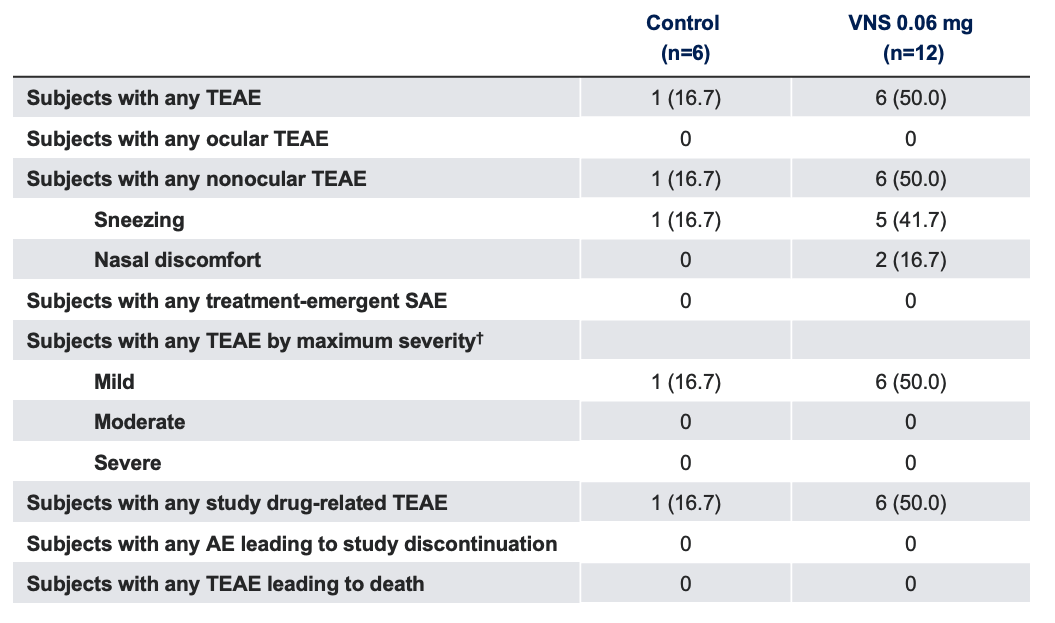
Note: The FDA-approved dosage of varenicline solution nasal spray is 0.03 mg BID.2 *Data presented as n (%); †Patients reporting more than one event were counted only once at the maximum severity reported.
AE, adverse event; FDA, Food and Drug Administration; SAE, serious adverse event; TEAE, treatment-emergent adverse event; VNS, varenicline solution nasal spray.
1. Dieckmann G.M. et al.Ophthalmol Ther. 2022;11:1551–1561; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.
AE, adverse event; FDA, Food and Drug Administration; SAE, serious adverse event; TEAE, treatment-emergent adverse event; VNS, varenicline solution nasal spray.
1. Dieckmann G.M. et al.Ophthalmol Ther. 2022;11:1551–1561; 2. TYRVAYA®(varenicline solution) nasal spray. Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/213978s005lbl.pdf.

