Single-center, Single-arm, Investigator-Initiated Trial to Evaluate the Efficacy of Varenicline Solution Nasal Spray on Cytokine Levels in the Tears of Subjects with Dry Eye Disease Associated with Moderate-to-Severe Sjogren’s Disease
| Molecule | Varenicline solution nasal spray (formerly known as OC-01) |
| Therapeutic focus | Dry eye disease |
| Phase | Phase 4 |
| Status | Completed |
| Participants | 39 |
| Completion Date | August 31, 2023 |
Note: This clinical trial was not included in the submission which resulted in Tyrvaya® approval.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.

Trial Design
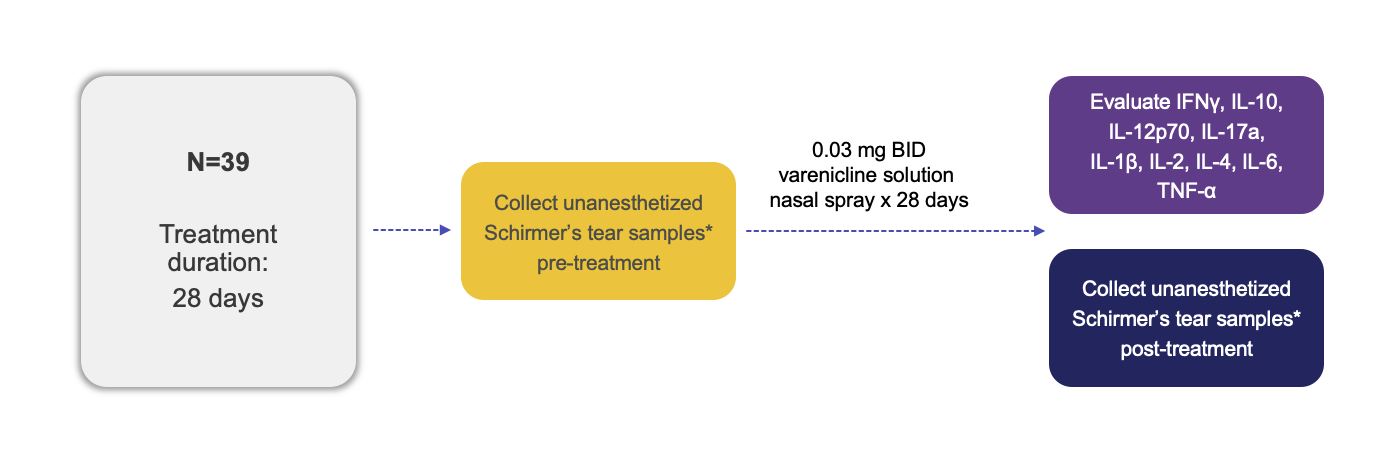
*A validated multiplex Luminex® bead-based immunoassay was used to analyze the Schirmer’s test strips at a lab. Luminex is a registered trademark of Luminex Corporation.
BID, twice daily; IFN, interferon; IL, interleukin; TNF, tumor necrosis factor.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
BID, twice daily; IFN, interferon; IL, interleukin; TNF, tumor necrosis factor.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
Baseline Characteristics
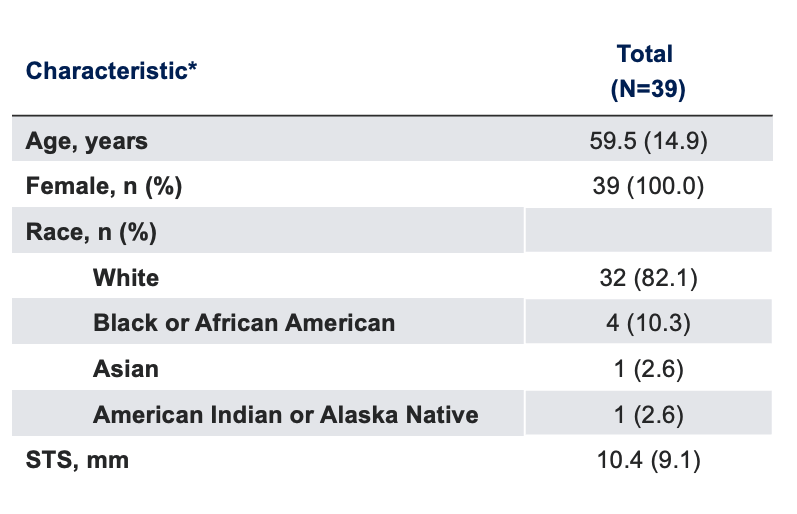
*Data presented as mean (SD), unless otherwise specified.
SD, standard deviation; STS, Schirmer’s test score.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
SD, standard deviation; STS, Schirmer’s test score.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
Efficacy Outcome Measures – Primary Endpoint
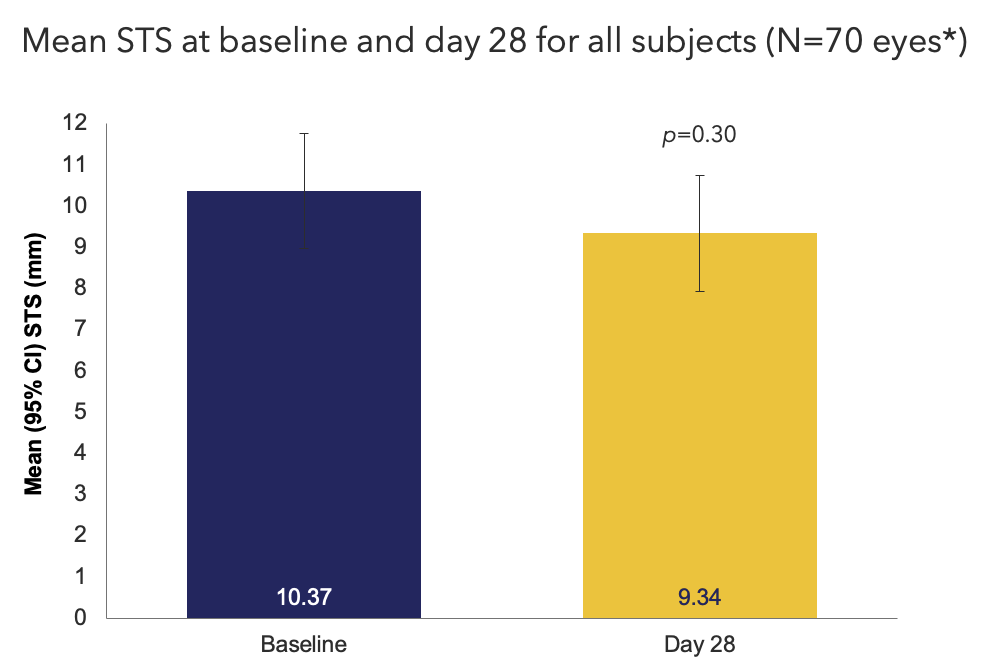
*3 subjects had baseline STS of 35 mm.
CI, confidence interval; STS, Schirmer's test score.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
CI, confidence interval; STS, Schirmer's test score.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
Efficacy Outcome Measures – Primary Endpoint
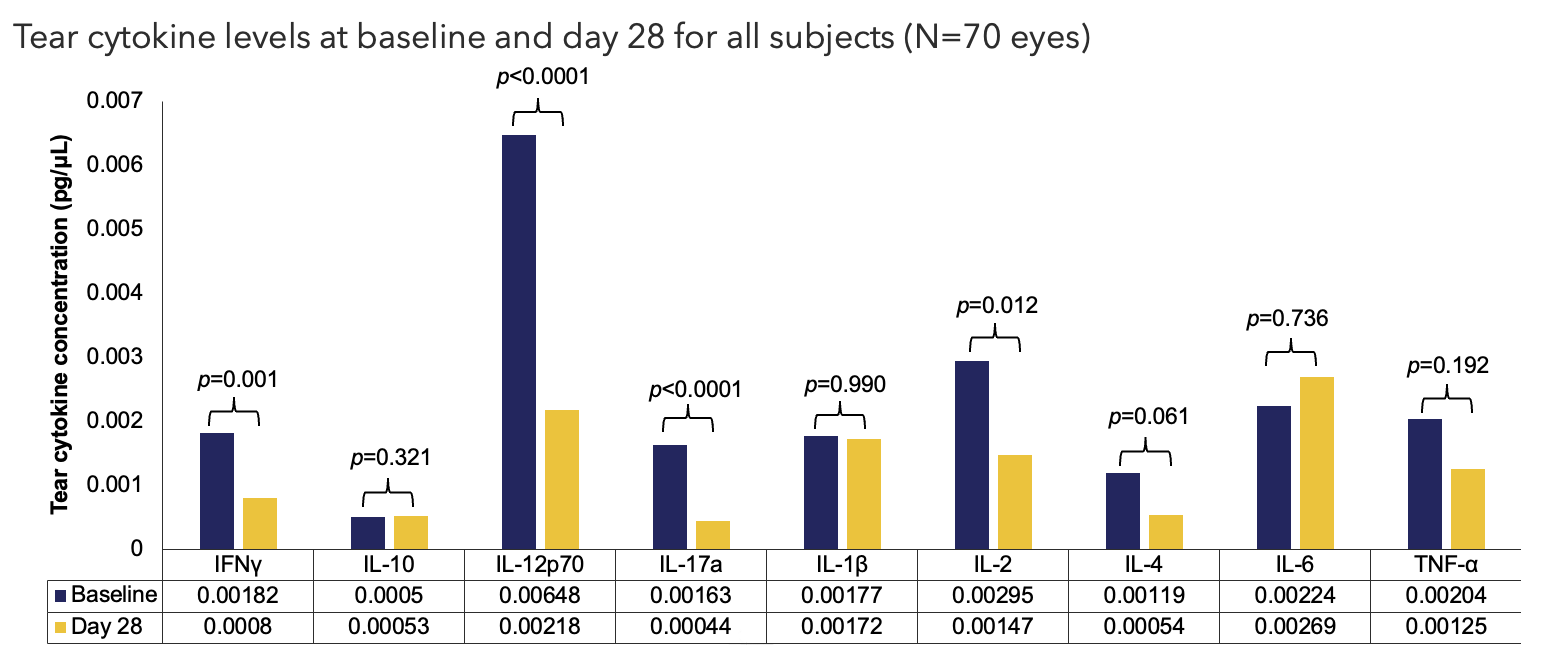
LLOQ values have been imputed by smallest value per parameter by visit (Baseline: IFNγ=0.000282243, IL-10=0.0004984466, IL-12p70=0.0002603448, IL-17a=0.0000257386, IL-1β=0.0002193732, IL-2=0.0002064163,
IL-4=0.0001430653, IL-6=0.0004244109, TNF-α=0.0001842957; Postbaseline: IFNγ=0.0002129771, IL-10=0.0004984466, IL-12p70=0.0000531067, IL-17a=0.0000649204, IL-1β=0.0000285413, IL-2=0.0014717635,
IL-4=0.0002345545, IL-6=0.0002425353, TNF-α=0.0001093103). One sample t-tests were used to generate p-values.
IFN, interferon; IL, interleukin; LLOQ, lower limit of quantitation; TNF, tumor necrosis factor.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
IL-4=0.0001430653, IL-6=0.0004244109, TNF-α=0.0001842957; Postbaseline: IFNγ=0.0002129771, IL-10=0.0004984466, IL-12p70=0.0000531067, IL-17a=0.0000649204, IL-1β=0.0000285413, IL-2=0.0014717635,
IL-4=0.0002345545, IL-6=0.0002425353, TNF-α=0.0001093103). One sample t-tests were used to generate p-values.
IFN, interferon; IL, interleukin; LLOQ, lower limit of quantitation; TNF, tumor necrosis factor.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
Efficacy Outcome Measures
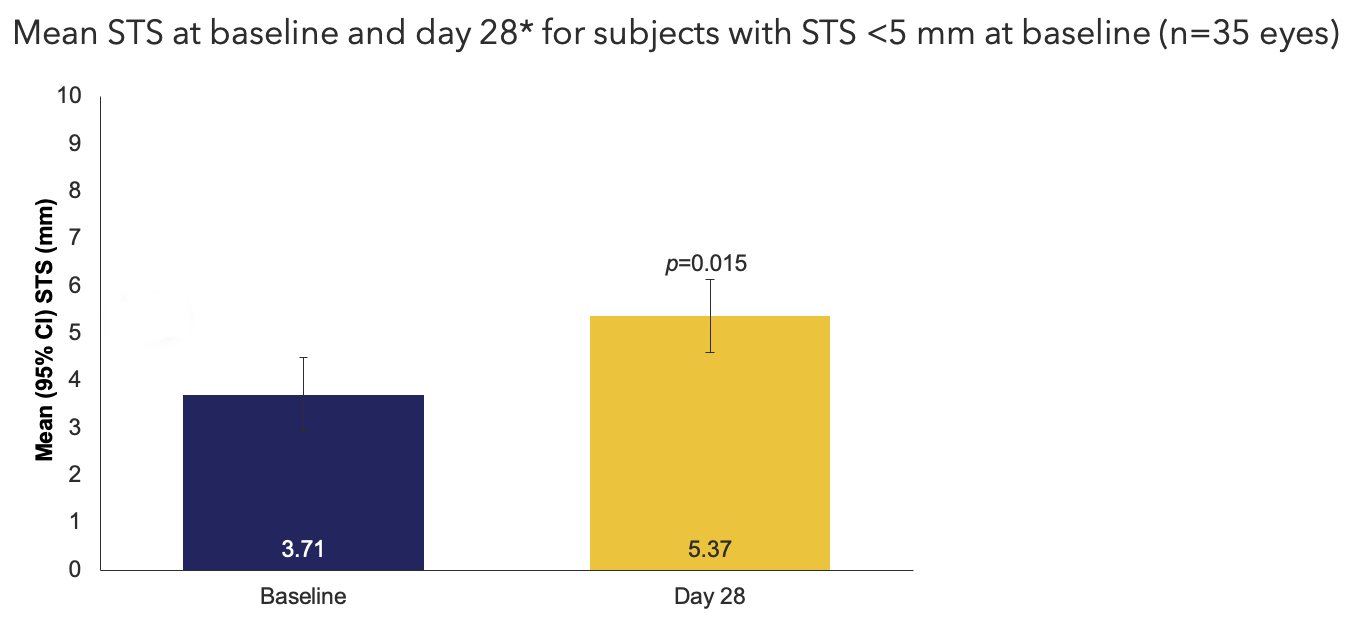
*Schirmer’s test was conducted within 4 hours of VNS use.
CI, confidence interval; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
CI, confidence interval; STS, Schirmer's test score; VNS, varenicline solution nasal spray.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
Efficacy Outcome Measures
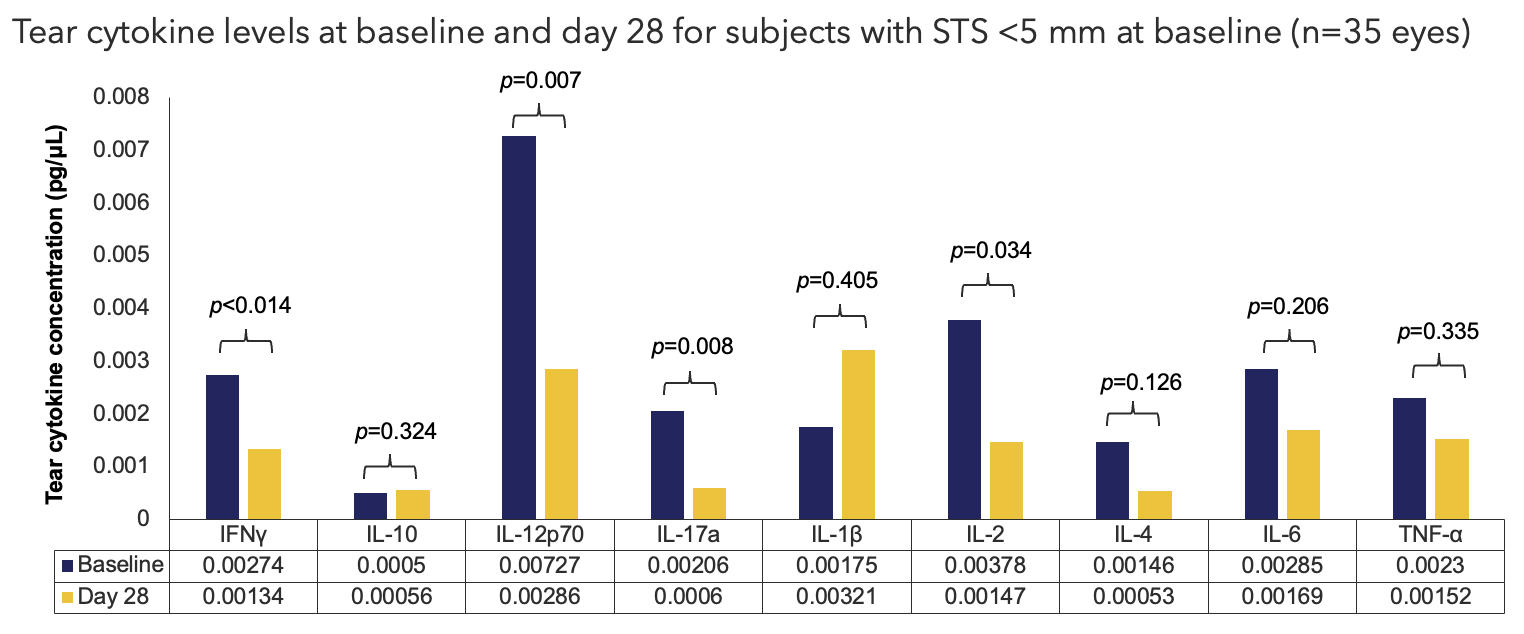
LLOQ values have been imputed by smallest value per parameter by visit (Baseline: IFNγ=0.000282243, IL-10=0.0004984466, IL-12p70=0.0002603448, IL-17a=0.0000257386, IL-1β=0.0002193732, IL-2=0.0002064163, IL-4=0.0001430653, IL-6=0.0004244109, TNF-α=0.0001842957; Postbaseline: IFNγ=0.0002129771, IL-10=0.0004984466, IL-12p70=0.0000531067, IL-17a=0.0000649204, IL-1β=0.0000285413, IL-2=0.0014717635, IL-4=0.0002345545, IL-6=0.0002425353, TNF-α=0.0001093103). One sample t-tests were used to generate p-values.
IFN, interferon; IL, interleukin; LLOQ, lower limit of quantitation; STS, Schirmer's test score; TNF, tumor necrosis factor.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.
IFN, interferon; IL, interleukin; LLOQ, lower limit of quantitation; STS, Schirmer's test score; TNF, tumor necrosis factor.
Cytokine Sub-Study Oyster Point Pharma, Inc. Data on file.

