Randomized, Parallel Arm, Double-Masked, Placebo-Controlled Study of the Safety and Efficacy of 0.75% Phentolamine Ophthalmic Solution to Reverse Pharmacologically Induced Mydriasis in Healthy Pediatric Subjects
| Molecule | Phentolamine ophthalmic solution 0.75% |
| Therapeutic focus | Reversal of pharmacologically-induced mydriasis |
| Phase | 3 |
| Status | Completed |
| Participants | 23 |
| Completion Date | April 28, 2022 |
https://clinicaltrials.gov/study/NCT05223478.

Trial Design1,2
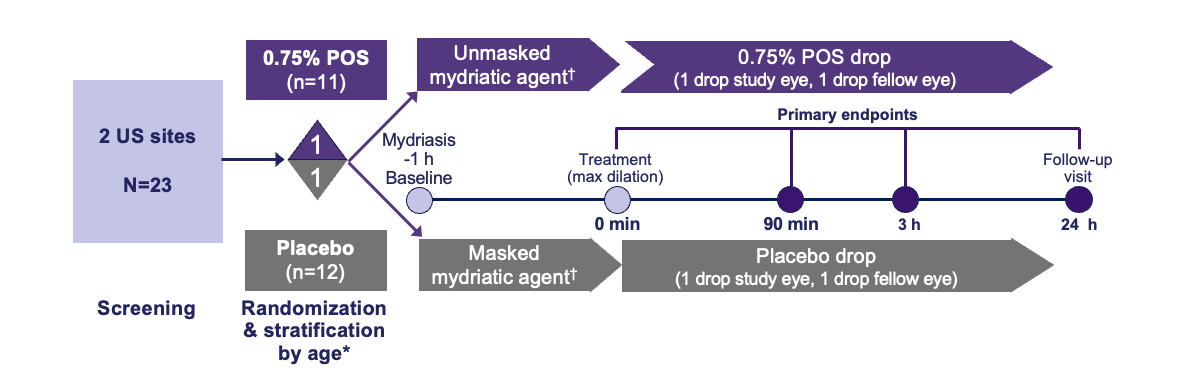
*Stratification was 1:1 between 3–5 and 6–11 years old; †Mydriatic agent randomization: 3:1:1. A: 2.5% phenylephrine;2 B: 1% tropicamide;2 C: Paremyd®†
(1% hydroxyamphetamine/0.25% tropicamide).3,4 †Paremyd is a registered trademark of Akorn, Inc.
h, hour; max, maximum; min, minute; POS, phentolamine ophthalmic solution; US, United States.
1. https://clinicaltrials.gov/study/NCT05223478; 2. MIRA-4 Ocuphire Pharma, Inc. Data on file; 3. RYZUMVI™ (phentolamine ophthalmic solution). Prescribing Information. Available at: https://www.ryzumvi.com/files/prescribing-information.pdf; 4. PAREMYD® (hydroxyamphetamine hydrobromide/ tropicamide ophthalmic solution). Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2001/19261s8lbl.pdf.
h, hour; max, maximum; min, minute; POS, phentolamine ophthalmic solution; US, United States.
1. https://clinicaltrials.gov/study/NCT05223478; 2. MIRA-4 Ocuphire Pharma, Inc. Data on file; 3. RYZUMVI™ (phentolamine ophthalmic solution). Prescribing Information. Available at: https://www.ryzumvi.com/files/prescribing-information.pdf; 4. PAREMYD® (hydroxyamphetamine hydrobromide/ tropicamide ophthalmic solution). Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2001/19261s8lbl.pdf.
Baseline Characteristics
 *Data presented as mean (SD), unless otherwise specified;1,2 †For BCDVA, the number of letters read is from the 4-meter distance only, so that 55 letters read is equivalent to a Snellen acuity of 20/20.2 ‡Paremyd is a registered trademark of Akorn, Inc.
*Data presented as mean (SD), unless otherwise specified;1,2 †For BCDVA, the number of letters read is from the 4-meter distance only, so that 55 letters read is equivalent to a Snellen acuity of 20/20.2 ‡Paremyd is a registered trademark of Akorn, Inc.BCDVA, best-corrected distance visual acuity; PD, pupil diameter; POS, phentolamine ophthalmic solution; SD, standard deviation.
1. https://clinicaltrials.gov/study/NCT05223478; 2. MIRA-4 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
 *The mITT population included all randomized subjects who received 1 drop of study medication in the study eye and then had at least 1 scheduled post-treatment PD measurement during visit 1 in the study eye.2
*The mITT population included all randomized subjects who received 1 drop of study medication in the study eye and then had at least 1 scheduled post-treatment PD measurement during visit 1 in the study eye.2mITT, modified intention-to-treat; PD, pupil diameter; POS, phentolamine ophthalmic solution.
1. https://clinicaltrials.gov/study/NCT05223478; 2. MIRA-4 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
 The mITT population included all randomized subjects who received 1 drop of study medication in the study eye and then had at least 1 scheduled post-treatment PD measurement during visit 1 in the study eye.2
The mITT population included all randomized subjects who received 1 drop of study medication in the study eye and then had at least 1 scheduled post-treatment PD measurement during visit 1 in the study eye.2mITT, modified Intention-to-Treat; PD, pupil diameter; POS, phentolamine ophthalmic solution; SD, standard deviation.
1. https://clinicaltrials.gov/study/NCT05223478; 2. MIRA-4 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
 The mITT population included all randomized subjects who received 1 drop of study medication in the study eye and then had at least 1 scheduled post-treatment PD measurement during visit 1 in the study eye.
The mITT population included all randomized subjects who received 1 drop of study medication in the study eye and then had at least 1 scheduled post-treatment PD measurement during visit 1 in the study eye.mITT, modified Intention-to-Treat; PD, pupil diameter; POS, phentolamine ophthalmic solution; SD, standard deviation.
MIRA-4 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
 The mITT population included all randomized subjects who received 1 drop of study medication in the study eye and then had at least 1 scheduled post-treatment PD measurement during visit 1 in the study eye.2
The mITT population included all randomized subjects who received 1 drop of study medication in the study eye and then had at least 1 scheduled post-treatment PD measurement during visit 1 in the study eye.2mITT, modified Intention-to-Treat; PD, pupil diameter; POS, phentolamine ophthalmic solution; SD, standard deviation.
1. https://clinicaltrials.gov/study/NCT05223478; 2. MIRA-4 Ocuphire Pharma, Inc. Data on file.
Safety Outcome Measures – Primary Endpoint
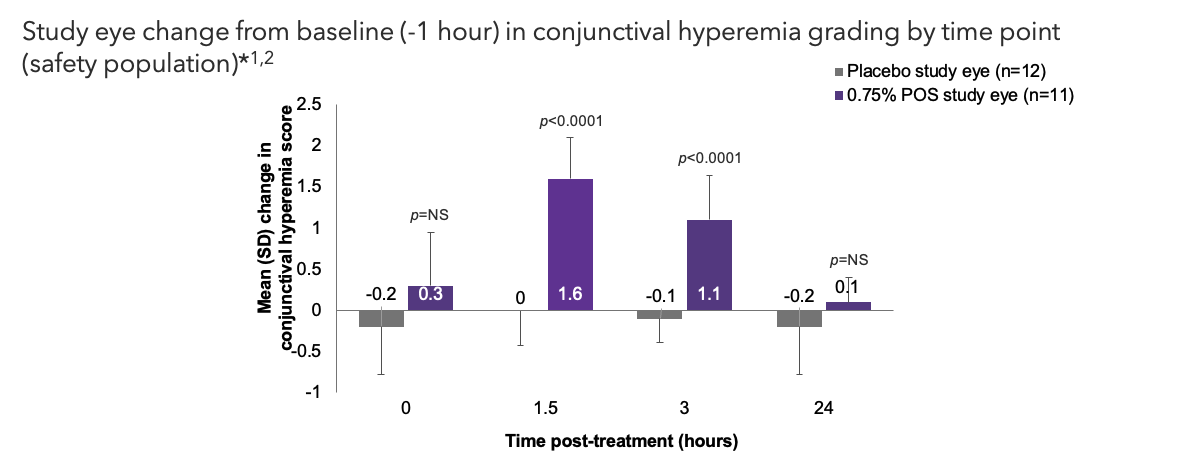
- At baseline (-1 hr), mean (SD) conjunctival hyperemia score was 0.4 (0.50) in study eyes treated with 0.75% POS and 0.8 (0.39) in study eyes treated with placebo2
CCLRU, Cornea and Contact Lens Research Unit; NS, not significant; POS, phentolamine ophthalmic solution; SD, standard deviation.
1. https://clinicaltrials.gov/study/NCT05223478 ; 2. MIRA-4 Ocuphire Pharma, Inc. Data on file.
Safety Outcome Measures – Primary Endpoint
 *Graded with a CCLRU card 4-point grading scale: None (0) = Normal. Appears white with a small number of conjunctival blood vessels easily observed; Mild (+1) = Prominent, pinkish-red color of both the bulbar and palpebral conjunctiva; Moderate (+2) = Bright, scarlet-red color of the bulbar and palpebral conjunctiva; Severe (+3) = Beefy-red with petechiae, dark red bulbar and palpebral conjunctiva with evidence of subconjunctival hemorrhage. The safety population included all randomized subjects who received at least 1 drop of study medication.
*Graded with a CCLRU card 4-point grading scale: None (0) = Normal. Appears white with a small number of conjunctival blood vessels easily observed; Mild (+1) = Prominent, pinkish-red color of both the bulbar and palpebral conjunctiva; Moderate (+2) = Bright, scarlet-red color of the bulbar and palpebral conjunctiva; Severe (+3) = Beefy-red with petechiae, dark red bulbar and palpebral conjunctiva with evidence of subconjunctival hemorrhage. The safety population included all randomized subjects who received at least 1 drop of study medication.CCLRU, Cornea and Contact Lens Research Unit; POS, phentolamine ophthalmic solution.
MIRA-4 Ocuphire Pharma, Inc. Data on file.
Safety Outcome Measures – Primary Endpoint
Change from baseline (-1 hour) in BCDVA and vital signs2
- BCDVA: One study eye in the POS 0.75% group lost ≥5 letters in BCDVA at 0 min; no other study eyes lost ≥5 letters in either treatment group at any time point
- Vital signs: Mean SBP, DBP, and HR were relatively unchanged, remained within normal limits throughout the duration of the study, and were similar between treatment groups
*Data presented as n (%).1
AE, adverse event; BCDVA, best-corrected distance visual acuity; DBP, diastolic blood pressure; HR, heart rate; POS, phentolamine ophthalmic solution; SAE, serious adverse event; SBP, systolic blood pressure.
1. https://clinicaltrials.gov/study/NCT05223478; 2. MIRA-4 Ocuphire Pharma, Inc. Data on file.
AE, adverse event; BCDVA, best-corrected distance visual acuity; DBP, diastolic blood pressure; HR, heart rate; POS, phentolamine ophthalmic solution; SAE, serious adverse event; SBP, systolic blood pressure.
1. https://clinicaltrials.gov/study/NCT05223478; 2. MIRA-4 Ocuphire Pharma, Inc. Data on file.

