Randomized, Parallel Arm, Double-Masked, Placebo-Controlled Study of the Safety and Efficacy of 0.75% Phentolamine Ophthalmic Solution to Reverse Pharmacologically-Induced Mydriasis in Healthy Subjects1
| Molecule | Phentolamine ophthalmic solution 0.75%1,2 |
| Therapeutic focus | Reversal of pharmacologically-induced mydriasis1,2 |
| Phase | 31,2 |
| Status | Completed1,2 |
| Participants | 3681,2 |
| Completion Date | March 29, 20221 |
1. https://clinicaltrials.gov/study/NCT05134974. 2. Pepose JS. et al. Ophthalmology. 2024.
doi: https://doi.org/10.1016/ j.ophtha.2024.09.010.

Trial Design1,2
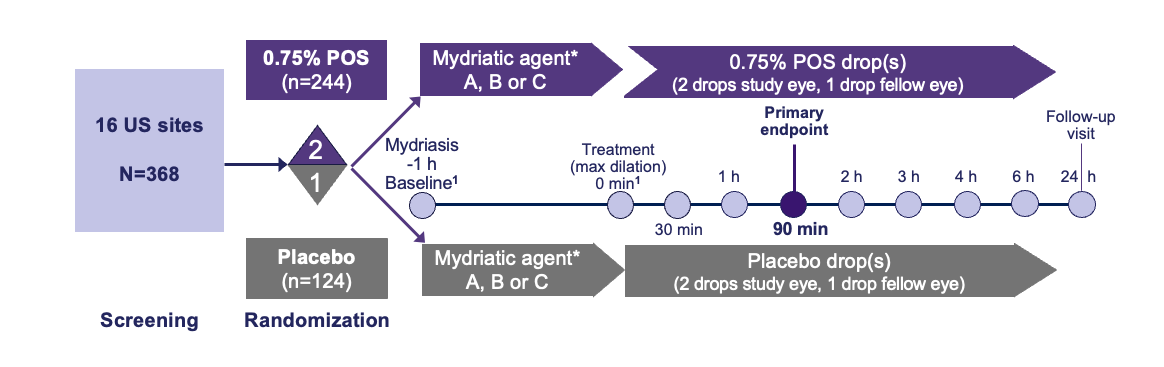
*Mydriatic agent randomization: 3:1:1. A: 2.5% phenylephrine;2 B: 1% tropicamide;2 C: Paremyd® †(1% hydroxyamphetamine/0.25% tropicamide).3,4
†Paremyd is a registered trademark of Akorn, Inc.
h, hour; max, maximum; min, minute; PD, pupil diameter; POS, phentolamine ophthalmic solution; US, United States.
1. https://clinicaltrials.gov/study/NCT05134974; 2. MIRA-3 Ocuphire Pharma, Inc. Data on file; 3. RYZUMVI™(phentolamine ophthalmic solution). Prescribing Information. Available at: https://www.ryzumvi.com/files/prescribing-information.pdf; 4. PAREMYD® (hydroxyamphetamine hydrobromide/ tropicamide ophthalmic solution). Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2001/19261s8lbl.pdf.
†Paremyd is a registered trademark of Akorn, Inc.
h, hour; max, maximum; min, minute; PD, pupil diameter; POS, phentolamine ophthalmic solution; US, United States.
1. https://clinicaltrials.gov/study/NCT05134974; 2. MIRA-3 Ocuphire Pharma, Inc. Data on file; 3. RYZUMVI™(phentolamine ophthalmic solution). Prescribing Information. Available at: https://www.ryzumvi.com/files/prescribing-information.pdf; 4. PAREMYD® (hydroxyamphetamine hydrobromide/ tropicamide ophthalmic solution). Prescribing Information. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2001/19261s8lbl.pdf.
Baseline Characteristics (1/2)
 *Data presented as mean (SD), unless otherwise specified;1,2 †Includes American Indian or Alaska Native, Native Hawaiian or other Pacific Islander, and unknown or not reported.1
*Data presented as mean (SD), unless otherwise specified;1,2 †Includes American Indian or Alaska Native, Native Hawaiian or other Pacific Islander, and unknown or not reported.1D, diopter; PD, pupil diameter; POS, phentolamine ophthalmic solution; SD, standard deviation.
1. https://clinicaltrials.gov/study/NCT05134974; 2. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Baseline Characteristics (2/2)
 *Data presented as mean (SD), unless otherwise specified;† The number of letters read is from the 4-meter distance only, so that 55 letters read is equivalent to a Snellen acuity of 20/20.
*Data presented as mean (SD), unless otherwise specified;† The number of letters read is from the 4-meter distance only, so that 55 letters read is equivalent to a Snellen acuity of 20/20.BCDVA, best-corrected distance visual acuity; DCNVA, distance-corrected near visual acuity; IOP, intraocular pressure; POS, phentolamine ophthalmic solution; SD, standard deviation.
MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Primary and Secondary Endpoints
 The mITT population included all randomized subjects who received 2 drops of study medication in the study eye and then had at least one scheduled PD measurement during visit 1. The mITT population was used for the primary endpoint analysis and to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.2
The mITT population included all randomized subjects who received 2 drops of study medication in the study eye and then had at least one scheduled PD measurement during visit 1. The mITT population was used for the primary endpoint analysis and to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.2 mITT, modified Intention-to-Treat; PD, pupil diameter; POS, phentolamine ophthalmic solution.
1. RYZUMVI™ (phentolamine ophthalmic solution). Prescribing Information. Available at: https://www.ryzumvi.com/files/prescribing-information.pdf; 2. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
 *p<0.01;1 The mITT population included all randomized subjects who received 2 drops of study medication in the study eye and then had at least one scheduled PD measurement during visit 1. The mITT population was used for the primary endpoint analysis and to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.2
*p<0.01;1 The mITT population included all randomized subjects who received 2 drops of study medication in the study eye and then had at least one scheduled PD measurement during visit 1. The mITT population was used for the primary endpoint analysis and to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.2mITT, modified Intention-to-Treat; PD, pupil diameter; POS, phentolamine ophthalmic solution.
1. RYZUMVI™ (phentolamine ophthalmic solution). Prescribing Information. Available at: https://www.ryzumvi.com/files/prescribing-information.pdf; 2. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
 *p<0.01;1 The mITT population included all randomized subjects who received 2 drops of study medication in the study eye and then had at least one scheduled PD measurement during visit 1. The mITT population was used for the primary endpoint analysis and to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.2
*p<0.01;1 The mITT population included all randomized subjects who received 2 drops of study medication in the study eye and then had at least one scheduled PD measurement during visit 1. The mITT population was used for the primary endpoint analysis and to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.2mITT, modified Intention-to-Treat; PD, pupil diameter; POS, phentolamine ophthalmic solution.
1. RYZUMVI™ (phentolamine ophthalmic solution). Prescribing Information. Available at: https://www.ryzumvi.com/files/prescribing-information.pdf; 2. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
 *Paremyd is a registered trademark of Akorn, Inc. †p<0.01;1 l The mITT population included all randomized subjects who received 2 drops of study medication in the study eye and then had at least one scheduled PD measurement during visit 1. The mITT population was used for the primary endpoint analysis and to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.2
*Paremyd is a registered trademark of Akorn, Inc. †p<0.01;1 l The mITT population included all randomized subjects who received 2 drops of study medication in the study eye and then had at least one scheduled PD measurement during visit 1. The mITT population was used for the primary endpoint analysis and to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.2mITT, modified Intention-to-Treat; PD, pupil diameter; POS, phentolamine ophthalmic solution.
1. RYZUMVI™ (phentolamine ophthalmic solution). Prescribing Information. Available at: https://www.ryzumvi.com/files/prescribing-information.pdf; 2. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
 The safety population included all randomized subjects who received at least 1 drop of study medication.
The safety population included all randomized subjects who received at least 1 drop of study medication.BCDVA, best-corrected distance visual acuity; POS, phentolamine ophthalmic solution.
MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
Percentage of subjects with unchanged accommodation from baseline (-1 hour) up to 6 hours (PP population)1
 The PP population included all subjects in the mITT population who had 2 drops of study medication in their study eye, had all scheduled PD measurements during visit 1, had an increase of >0.2 mm in PD in the study eye at time 0 min compared to baseline (-1 hr), and had no major protocol deviations. The PP population was used to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.1
The PP population included all subjects in the mITT population who had 2 drops of study medication in their study eye, had all scheduled PD measurements during visit 1, had an increase of >0.2 mm in PD in the study eye at time 0 min compared to baseline (-1 hr), and had no major protocol deviations. The PP population was used to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.1mITT, modified Intention-to-Treat; PP, per-protocol; PD, pupil diameter; POS, phentolamine ophthalmic solution 0.75%.1. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
Percentage of subjects with unchanged accommodation from baseline (-1 hour) up to 6 hours (PP population)1
 The PP population included all subjects in the mITT population who had 2 drops of study medication in their study eye, had all scheduled PD measurements during visit 1, had an increase of >0.2 mm in PD in the study eye at time 0 min compared to baseline (-1 hr), and had no major protocol deviations. The PP population was used to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.1
The PP population included all subjects in the mITT population who had 2 drops of study medication in their study eye, had all scheduled PD measurements during visit 1, had an increase of >0.2 mm in PD in the study eye at time 0 min compared to baseline (-1 hr), and had no major protocol deviations. The PP population was used to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.1mITT, modified Intention-to-Treat; PP, per-protocol; PD, pupil diameter; POS, phentolamine ophthalmic solution 0.75%.1. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
Mean Change in Accommodation in Study Eyes Receiving Phenylephrine by Time Point (PP population)1
 The PP population included all subjects in the mITT population who had 2 drops of study medication in their study eye, had all scheduled PD measurements during visit 1, had an increase of >0.2 mm in PD in the study eye at time 0 min compared to baseline (-1 hr), and had no major protocol deviations. The PP population was used to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.1
The PP population included all subjects in the mITT population who had 2 drops of study medication in their study eye, had all scheduled PD measurements during visit 1, had an increase of >0.2 mm in PD in the study eye at time 0 min compared to baseline (-1 hr), and had no major protocol deviations. The PP population was used to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.1mITT, modified Intention-to-Treat; PP, per-protocol; PD, pupil diameter; POS, phentolamine ophthalmic solution 0.75%.1. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
Mean Change in Accommodation in Study Eyes Receiving Tropicamide or Paramyd by Time Point (PP population)1
 The PP population included all subjects in the mITT population who had 2 drops of study medication in their study eye, had all scheduled PD measurements during visit 1, had an increase of >0.2 mm in PD in the study eye at time 0 min compared to baseline (-1 hr), and had no major protocol deviations. The PP population was used to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.1
The PP population included all subjects in the mITT population who had 2 drops of study medication in their study eye, had all scheduled PD measurements during visit 1, had an increase of >0.2 mm in PD in the study eye at time 0 min compared to baseline (-1 hr), and had no major protocol deviations. The PP population was used to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.1mITT, modified Intention-to-Treat; PP, per-protocol; PD, pupil diameter; POS, phentolamine ophthalmic solution 0.75.1. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
Change in BCDVA under glare conditions and under normal photopic lighting versus glare conditions1
- No significant differences in BCDVA were observed between Ryzumvi and placebo treatment at any time point when measuring BCDVA under glare or photopic lighting minus glare conditions.
Change in glare discomfort from maximum PD1
- Glare discomfort was not significantly different between Ryzumvi and placebo treated eyes.
1. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Secondary Endpoints
Mean Change in Pupillary Light Reflex in Study Eyes From 0 Min by Time Point (PP Population)1
 The PP population included all subjects in the mITT population who had 2 drops of study medication in their study eye, had all scheduled PD measurements during visit 1, had an increase of >0.2 mm in PD in the study eye at time 0 min compared to baseline (-1 hr), and had no major protocol deviations. The PP population was used to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.1
The PP population included all subjects in the mITT population who had 2 drops of study medication in their study eye, had all scheduled PD measurements during visit 1, had an increase of >0.2 mm in PD in the study eye at time 0 min compared to baseline (-1 hr), and had no major protocol deviations. The PP population was used to analyze selected secondary efficacy endpoints, with subjects included in their randomized treatment regardless of the treatment they actually received.1mITT, modified Intention-to-Treat; PP, per-protocol; PD, pupil diameter; POS, phentolamine ophthalmic solution 0.75.
1. MIRA-3 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcomes Measures- Secondary Endpoints
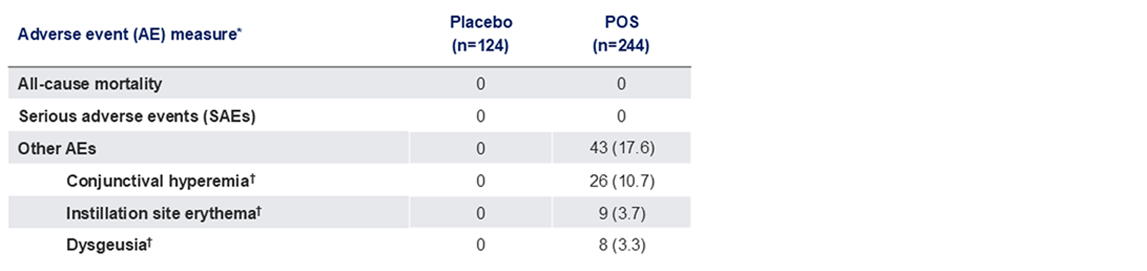
*Data presented as n (%); †Collected by systematic assessment.
POS, phentolamine ophthalmic solution.
https://clinicaltrials.gov/study/NCT05134974.
POS, phentolamine ophthalmic solution.
https://clinicaltrials.gov/study/NCT05134974.

