A Randomized, Double-Masked, Vehicle-Controlled, Phase 3 Study to Evaluate the Efficacy and Safety of Pimecrolimus 0.3% Ophthalmic Ointment (MR-139), in Subjects With Blepharitis
| Molecule | Pimecrolimus 0.3% Ophthalmic Ointment (MR-139) |
| Therapeutic focus | Blepharitis |
| Phase | 3 |
| Status | Enrolling |
| Participants | ~470 |
| Start Date | April 22, 2024 |
The use of MR-139 for Blepharitis is currently being investigated and its safety and efficacy have not been established.
https://clinicaltrials.gov/study/NCT06400511.

Trial Design
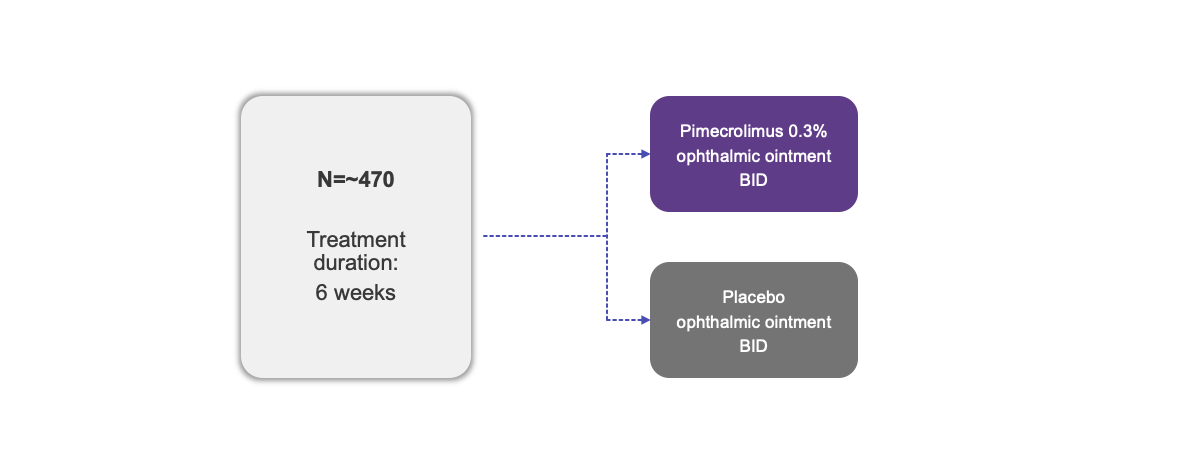
The use of MR-139 for Blepharitis is currently being investigated and its safety and efficacy have not been established.
BID, twice daily.
https://clinicaltrials.gov/study/NCT06400511.
Secondary Endpoints
- Change in ocular discomfort score (ODS)*,† from baseline to week 6
- Complete resolution (score 0 on the grading scale of 0–4†) of eyelid debris at week 12
- Change in ODS*,† from baseline to week 12
The use of MR-139 for Blepharitis is currently being investigated and its safety and efficacy have not been established.
*On a 100-mm visual analog scale from 0 (no discomfort) to 100 (maximal discomfort); †A higher score indicates a worse outcome.
ODS, ocular discomfort score.
https://clinicaltrials.gov/study/NCT06400511.
Safety Measurements
- Urine pregnancy test (if applicable, for women of childbearing potential)
- Slitlamp examination
- Corneal fluorescein staining (CFS)
- Conjunctival staining
- Intraocular pressure (IOP)
- Corrected distance visual acuity (CDVA)
- Adverse event (AE) assessment
- Dilated fundus exam at baseline and last visit
Famy Life Sciences, a Viatris Company. Data on file.

