Safety and Efficacy of 0.75% Phentolamine Ophthalmic Solution With Pilocarpine Eye Drops in Subjects With Presbyopia
| Molecule | Phentolamine ophthalmic solution 0.75% |
| Therapeutic focus | Presbyopia |
| Phase | 2 |
| Status | Completed |
| Participants | 150 |
| Completion Date | June 30, 2021 |
The use of MR-141 for Presbyopia is currently being investigated and its safety and efficacy have not been established.
https://clinicaltrials.gov/study/NCT04675151.

Trial Design1,2
 The use of MR-141 for Presbyopia is currently under investigation and its safety and efficacy have not been established.
The use of MR-141 for Presbyopia is currently under investigation and its safety and efficacy have not been established.LDP, low-dose pilocarpine; POS, phentolamine ophthalmic solution 0.75%. 1. https://clinicaltrials.gov/study/NCT04675151; 2. Pepose J.S., et al. ASCRS 2021 Presentation (Paper #76645).
Available at: https://d1io3yog0oux5.cloudfront.net/_024aeac324ab6f96d110f6a3fb5dd959/ocuphire/db/357/3653/pdf/ASCRS+2021+VEGA-1+Presentation_FINAL.pdf
Baseline Characteristics
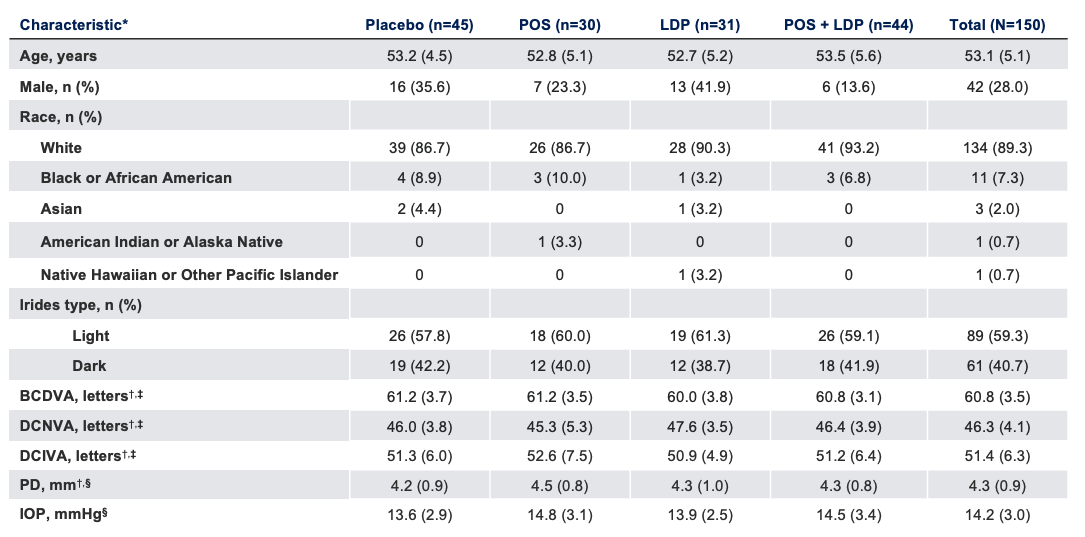
The use of MR-141 for Presbyopia is currently being investigated and its safety and efficacy have not been established.
*Data presented as mean (SD), unless otherwise specified; †Photopic; ‡Binocular; §Study eye.
BCDVA, best-corrected distance visual acuity; DCIVA, distance-corrected intermediate visual acuity; DCNVA, distance-corrected near visual acuity; IOP, intraocular pressure; LDP, low-dose pilocarpine; PD, pupil diameter; POS, phentolamine ophthalmic solution; SD, standard deviation.
VEGA-1 Ocuphire Pharma, Inc. Data on file.
*Data presented as mean (SD), unless otherwise specified; †Photopic; ‡Binocular; §Study eye.
BCDVA, best-corrected distance visual acuity; DCIVA, distance-corrected intermediate visual acuity; DCNVA, distance-corrected near visual acuity; IOP, intraocular pressure; LDP, low-dose pilocarpine; PD, pupil diameter; POS, phentolamine ophthalmic solution; SD, standard deviation.
VEGA-1 Ocuphire Pharma, Inc. Data on file.
Percent of Subjects With Improvement ≥ 15 Letters From Baseline in Photopic Binocular DCNVA by Time Point 1,2
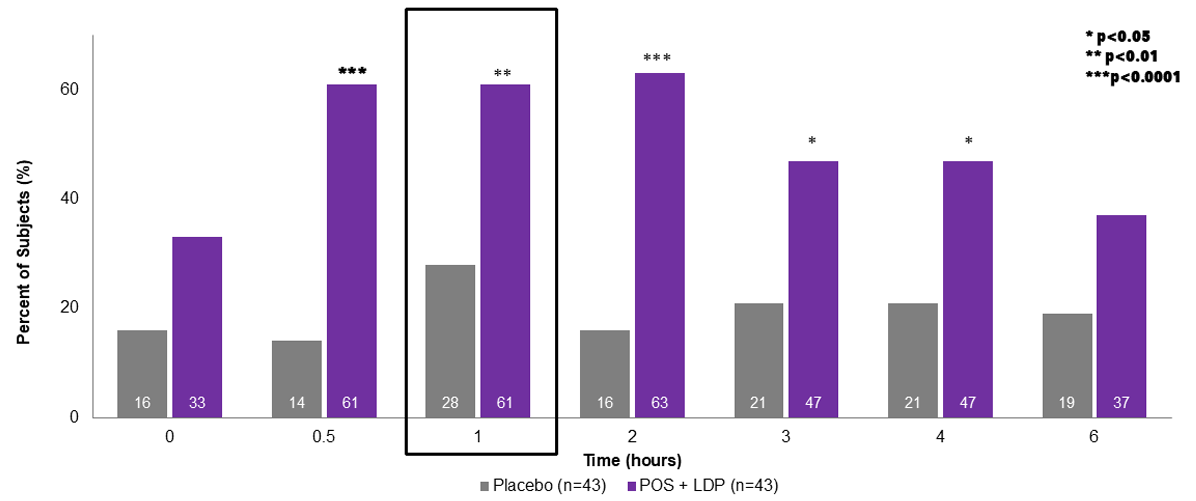
The use of MR-141 for Presbyopia is currently being investigated and its safety and efficacy have not been established.
DCNVA, distance-corrected near visual acuity; POS, phentolamine ophthalmic solution 0.75%; LDP, low dose pilocarpine
1. https://clinicaltrials.gov/study/NCT04675151. 2. Pepose J.S., et al. ASCRS 2021 Presentation (Paper #76645).
Available at: https://d1io3yog0oux5.cloudfront.net/_024aeac324ab6f96d110f6a3fb5dd959/ocuphire/db/357/3653/pdf/ASCRS+2021+VEGA-1+Presentation_FINAL.pdf.
DCNVA, distance-corrected near visual acuity; POS, phentolamine ophthalmic solution 0.75%; LDP, low dose pilocarpine
1. https://clinicaltrials.gov/study/NCT04675151. 2. Pepose J.S., et al. ASCRS 2021 Presentation (Paper #76645).
Available at: https://d1io3yog0oux5.cloudfront.net/_024aeac324ab6f96d110f6a3fb5dd959/ocuphire/db/357/3653/pdf/ASCRS+2021+VEGA-1+Presentation_FINAL.pdf.
Percent of Subjects With Improvement ≥ 15 Letters From Baseline in Photopic Binocular DCNVA And < 5 Letter Loss in Photopic BCDVA by Time Point 1,2
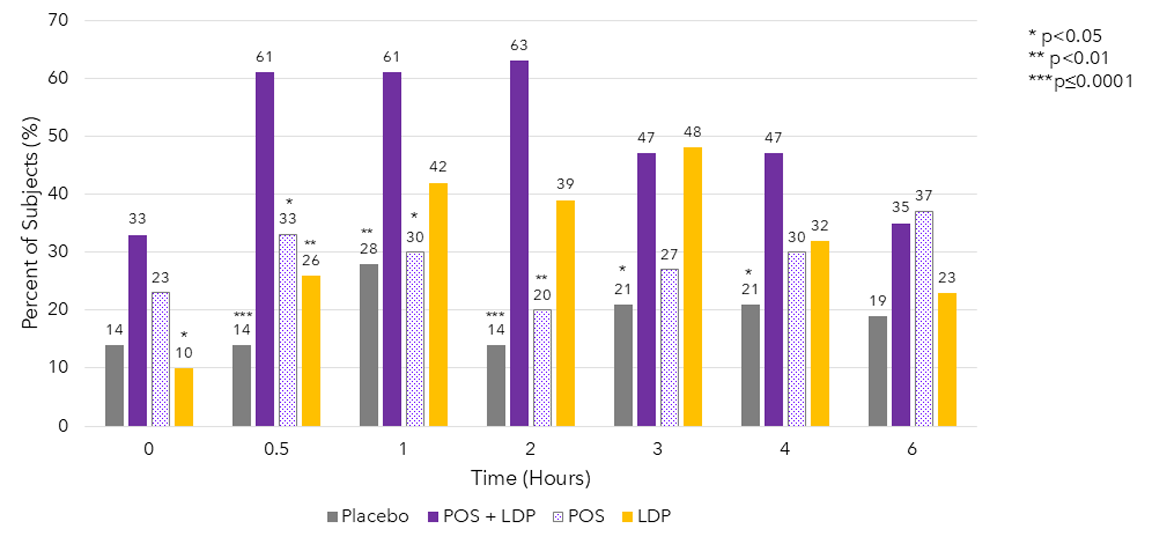
The use of MR-141 for Presbyopia is currently under investigation and its safety and efficacy have not been established.
BCDVA, best-corrected distance visual acuity; DCNVA, distance-corrected near visual acuity; LDP, low-dose pilocarpine; POS, phentolamine ophthalmic solution 0.75%. https://clinicaltrials.gov/study/NCT04675151. 2. Pepose J.S., et al. ASCRS 2021 Presentation (Paper #76645).
Available at: https://d1io3yog0oux5.cloudfront.net/_024aeac324ab6f96d110f6a3fb5dd959/ocuphire/db/357/3653/pdf/ASCRS+2021+VEGA-1+Presentation_FINAL.pdf.
BCDVA, best-corrected distance visual acuity; DCNVA, distance-corrected near visual acuity; LDP, low-dose pilocarpine; POS, phentolamine ophthalmic solution 0.75%. https://clinicaltrials.gov/study/NCT04675151. 2. Pepose J.S., et al. ASCRS 2021 Presentation (Paper #76645).
Available at: https://d1io3yog0oux5.cloudfront.net/_024aeac324ab6f96d110f6a3fb5dd959/ocuphire/db/357/3653/pdf/ASCRS+2021+VEGA-1+Presentation_FINAL.pdf.
Mean Photopic Pupil Diameter in Best Eye by Time Point1,2
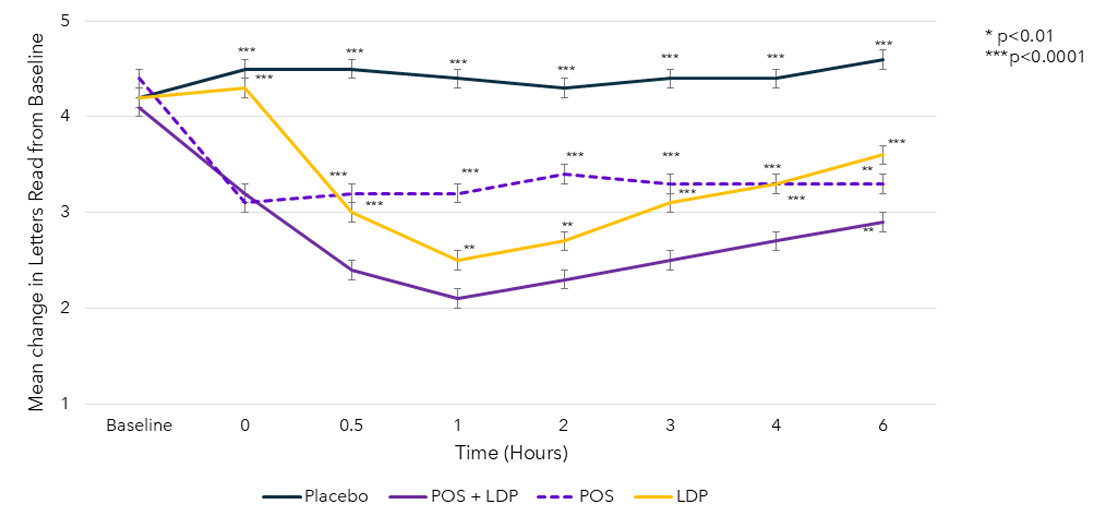
The use of MR-141 for Presbyopia is currently under investigation and its safety and efficacy have not been established.
BCDVA, best-corrected distance visual acuity; DCNVA, distance-corrected near visual acuity; LDP, low-dose pilocarpine; POS, phentolamine ophthalmic solution 0.75%. https://clinicaltrials.gov/study/NCT04675151. 2. Pepose J.S., et al. ASCRS 2021 Presentation (Paper #76645).
Available at: https://d1io3yog0oux5.cloudfront.net/_024aeac324ab6f96d110f6a3fb5dd959/ocuphire/db/357/3653/pdf/ASCRS+2021+VEGA-1+Presentation_FINAL.pdf.
BCDVA, best-corrected distance visual acuity; DCNVA, distance-corrected near visual acuity; LDP, low-dose pilocarpine; POS, phentolamine ophthalmic solution 0.75%. https://clinicaltrials.gov/study/NCT04675151. 2. Pepose J.S., et al. ASCRS 2021 Presentation (Paper #76645).
Available at: https://d1io3yog0oux5.cloudfront.net/_024aeac324ab6f96d110f6a3fb5dd959/ocuphire/db/357/3653/pdf/ASCRS+2021+VEGA-1+Presentation_FINAL.pdf.
Safety Outcome Measures*
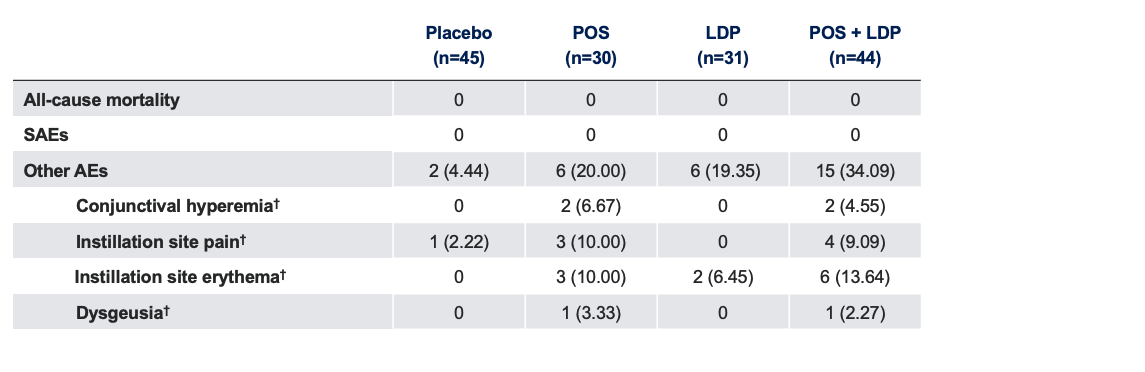
The use of MR-141 for Presbyopia is currently being investigated and its safety and efficacy have not been established.
*Data presented as n (%); †Collected by systematic assessment.
AE, adverse event; LDP, low-dose pilocarpine; POS, phentolamine ophthalmic solution; SAE, serious adverse event.
https://clinicaltrials.gov/study/NCT04675151.
*Data presented as n (%); †Collected by systematic assessment.
AE, adverse event; LDP, low-dose pilocarpine; POS, phentolamine ophthalmic solution; SAE, serious adverse event.
https://clinicaltrials.gov/study/NCT04675151.

