Randomized, Placebo-Controlled, Double-Masked Study of the Safety and Efficacy of POS (0.75% Phentolamine Ophthalmic Solution) in Subjects Who Have Previously Had Keratorefractive Surgery and Have Decreased Visual Acuity Under Mesopic Conditions
| Molecule | Phentolamine ophthalmic solution 0.75% |
| Therapeutic focus | Dim light disturbances |
| Phase | 3 |
| Status | Ongoing |
| Participants | 200 |
| Start Date | April 1, 2024 |
The use of MR-142 for Dim Light Disturbances is currently being investigated and its safety and efficacy have not been established.
https://clinicaltrials.gov/study/NCT06349759.

Trial Design1
 The use of MR-142 for Dim Light Disturbances is currently being investigated and its safety and efficacy have not been established.
The use of MR-142 for Dim Light Disturbances is currently being investigated and its safety and efficacy have not been established.All subjects will have safety and efficacy assessments at each office visit during stage 1; all subjects will have safety assessments at each office visit during optional stage 2.
*Each subject stratified by iris color (light/dark irides); †mLCVA evaluations performed on these days.
mLCVA, mesopic low-contrast distance visual acuity; POS, phentolamine ophthalmic solution; QD, once daily; US, United States.
LYNX-2 Ocuphire Pharma, Inc. Data on file.
Key Secondary Endpoints1
- Percentage of subjects with ≥ 10 and ≥ 15 ETDRS letters improvement in mLCVA compared to Baseline (Day 1 pre-dose) at Day 8
- Mean and change from Baseline (Day 1 pre-dose) in mLCVA and mHCVA at Days 3, 8, and 15 and Week 6
ETDRS, Early Treatment Diabetic Retinopathy Study; mHCVA, mesopic high-contrast distance visual acuity; mLCVA, mesopic low contrast visual acuity.
https://clinicaltrials.gov/study/NCT06349759 1. Chu et al. ASCRS 2026 (Paper # 119714)
Baseline Characteristics
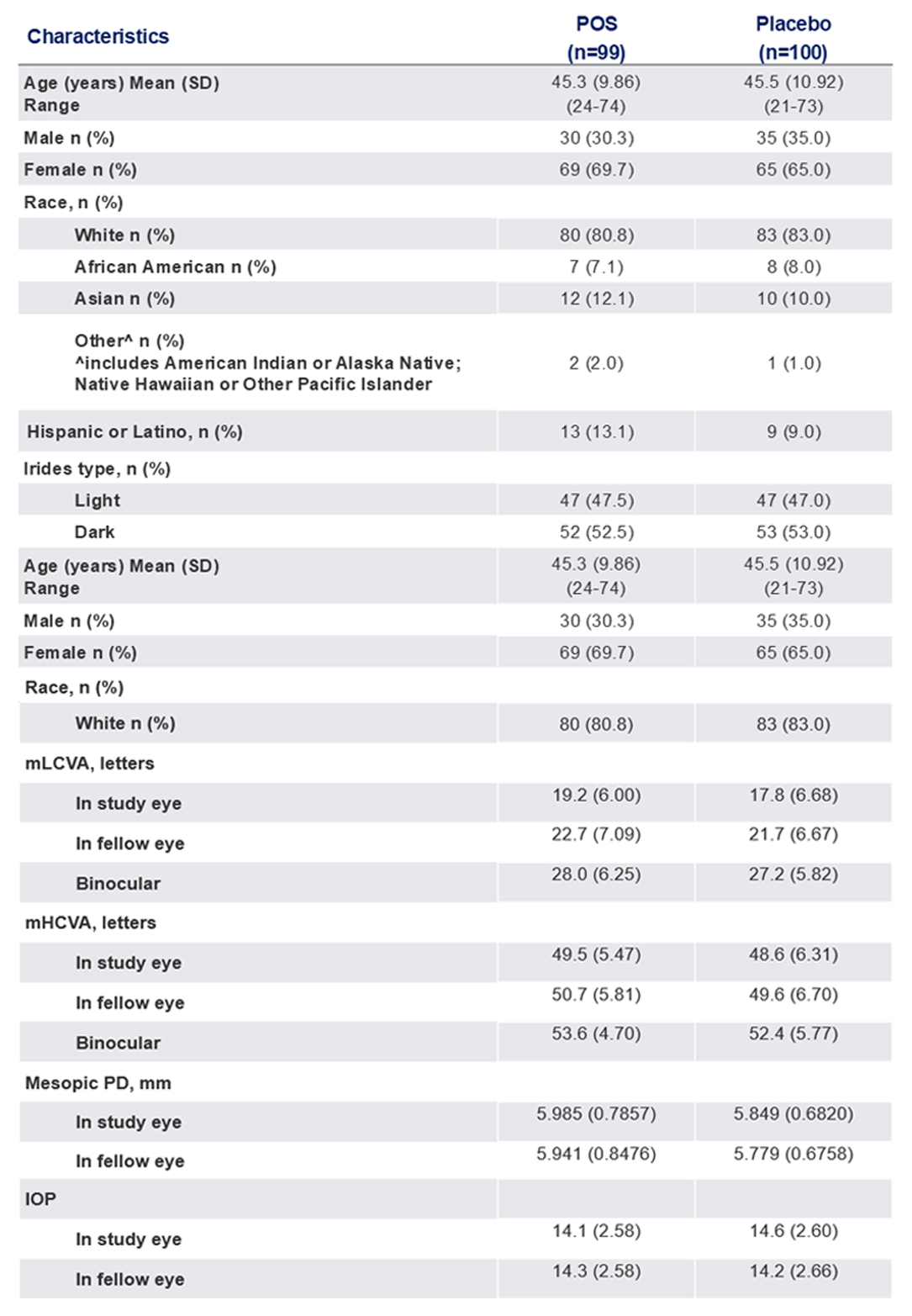
The use of MR-142 for Dim Light Disturbances is currently being investigated and its safety and efficacy have not been established.
POS, phentolamine ophthalmic solution 0.75%; mLCVA, mesopic low-contrast distance visual acuity; mHCVA, mesopic high-contrast distance visual acuity; IOP, intraocular pressure; PD, pupil diameter
https://clinicaltrials.gov/study/NCT06349759. LYNX-2 Ocuphire Pharma, Inc. Data on file.
POS, phentolamine ophthalmic solution 0.75%; mLCVA, mesopic low-contrast distance visual acuity; mHCVA, mesopic high-contrast distance visual acuity; IOP, intraocular pressure; PD, pupil diameter
https://clinicaltrials.gov/study/NCT06349759. LYNX-2 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Primary and Secondary Endpoints
Percentage of Subjects Gaining ≥15 Letters mLCVA at Day 15 1
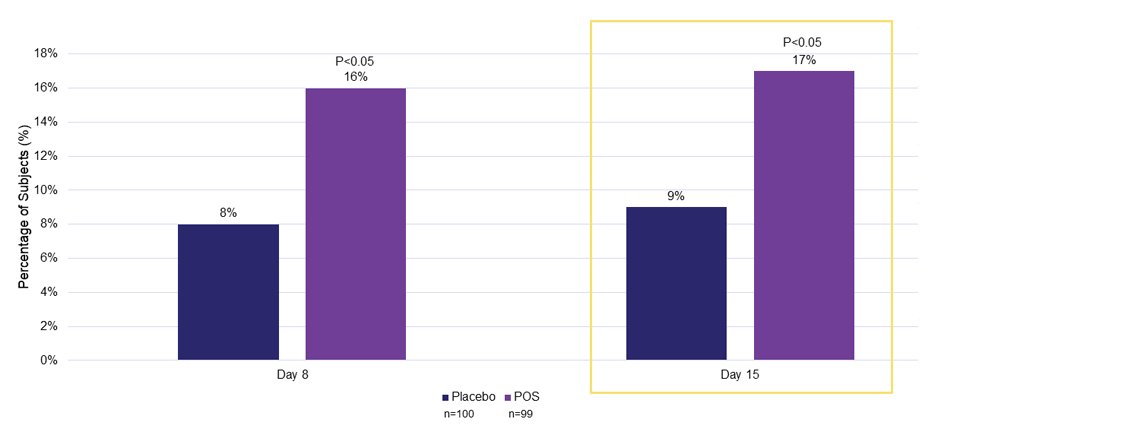
The use of MR-142 for Dim Light Disturbances is currently being investigated and its safety and efficacy have not been established.
POS, phentolamine ophthalmic solution 0.75%; mLCVA, mesopic low-contrast visual acuity.
https://clinicaltrials.gov/study/NCT06349759 1. Chu et al. ASCRS 2026 (Paper # 119714)
POS, phentolamine ophthalmic solution 0.75%; mLCVA, mesopic low-contrast visual acuity.
https://clinicaltrials.gov/study/NCT06349759 1. Chu et al. ASCRS 2026 (Paper # 119714)
Efficacy Outcome Measures – Secondary Endpoint
Change from Baseline in mLCVA at Week 6 vs Visits Through Day 15 in Phentolamine Treated Study Eyes1
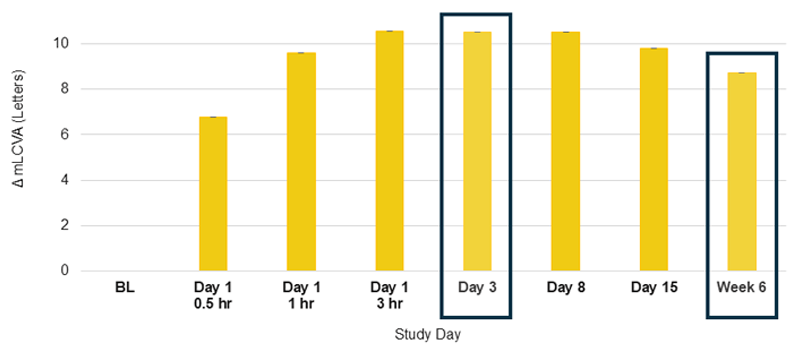
The use of MR-142 for Dim Light Disturbances is currently being investigated and its safety and efficacy have not been established.
Phentolamine, phentolamine ophthalmic solution 0.75%; mLCVA, mesopic low-contrast visual acuity.
https://clinicaltrials.gov/study/NCT06349759 1. Chu et al. ASCRS 2026 (Paper # 119714)
Phentolamine, phentolamine ophthalmic solution 0.75%; mLCVA, mesopic low-contrast visual acuity.
https://clinicaltrials.gov/study/NCT06349759 1. Chu et al. ASCRS 2026 (Paper # 119714)
Safety Outcome Measures
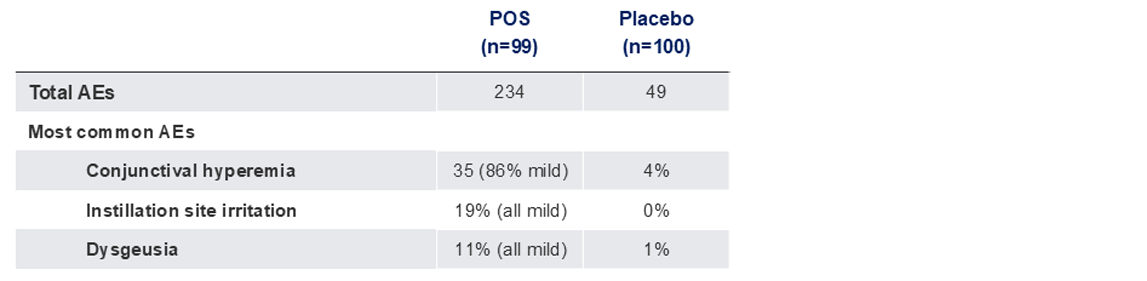
The use of MR-142 for Dim Light Disturbances is currently being investigated and its safety and efficacy have not been established.
TEAEs, Treatment-related adverse events; AE, adverse event; POS, phentolamine ophthalmic solution 0.75%.
https://clinicaltrials.gov/study/NCT06349759 1. Chu et al. ASCRS 2026 (Paper # 119714)
TEAEs, Treatment-related adverse events; AE, adverse event; POS, phentolamine ophthalmic solution 0.75%.
https://clinicaltrials.gov/study/NCT06349759 1. Chu et al. ASCRS 2026 (Paper # 119714)

