Randomized, Placebo-Controlled, Double-Masked Study of the Safety and Efficacy of 0.75% Phentolamine Ophthalmic Solution in Subjects With Dim Light Vision Disturbances1
| Molecule | Phentolamine ophthalmic solution 0.75%1 |
| Therapeutic focus | Dim light disturbances1 |
| Phase | 31 |
| Status | Completed1 |
| Participants | 1452 |
| Completion Date | May 19, 20221 |
The use of MR-142 for Dim Light Disturbances is currently being investigated and its safety and efficacy have not been established.
1. https://clinicaltrials.gov/study/NCT04638660; 2. LYNX-1 Ocuphire Pharma, Inc. Data on file.

Trial Design1
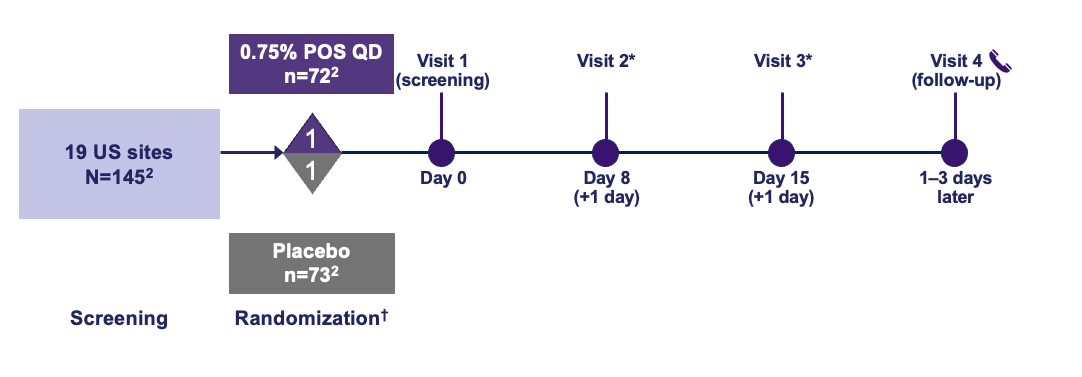
The use of MR-142 for Dim Light Disturbances is currently being investigated and its safety and efficacy have not been established.
At select sites, OPD-Scan measurements were made using wavefront aberrometry. *mLCVA evaluations performed on these days; †Each subject stratified by iris color (light/dark irides).
mLCVA, mesopic low-contrast distance visual acuity; OPD, optical path difference; POS, phentolamine ophthalmic solution; OD, once daily.
1. https://clinicaltrials.gov/study/NCT04638660; 2. LYNX-1 Ocuphire Pharma, Inc. Data on file.
Baseline Characteristics

*Data presented as mean (SD), unless otherwise specified.1,2 DCNVA, distance-corrected near visual acuity; mLCVA, mesopic low-contrast distance visual acuity; PD, pupil diameter; pLCVA, photopic low-contrast distance visual acuity; POS, phentolamine ophthalmic solution; SD, standard deviation.
1. https://clinicaltrials.gov/study/NCT04638660; 2. LYNX-1 Ocuphire Pharma, Inc. Data on file.
Efficacy Outcome Measures – Primary and Secondary Endpoints
Percentage of subjects with ≥15 ETDRS letters (≥3 lines) of improvement in the study eye in monocular mLCVA from baseline to days 8 and 151,2
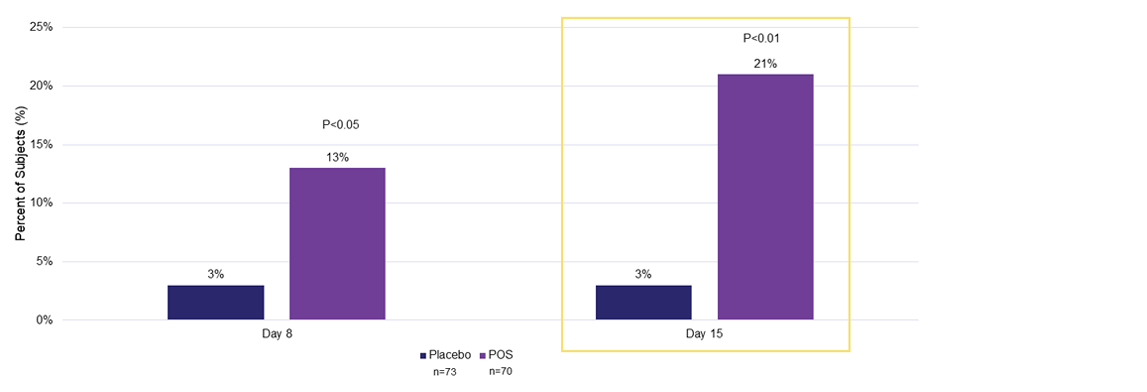
ETDRS, Early Treatment Diabetic Retinopathy Study; mLCVA, mesopic low-contrast distance visual acuity; POS, phentolamine ophthalmic solution.
1. LYNX-1 Ocuphire Pharma, Inc. Data on file; 2. Pepose JS, et al. ARVO 2023 Presentation Abstract (#2506). Available at: https://iovs.arvojournals.org/article.aspx?articleid=2790304
Safety Outcome Measures
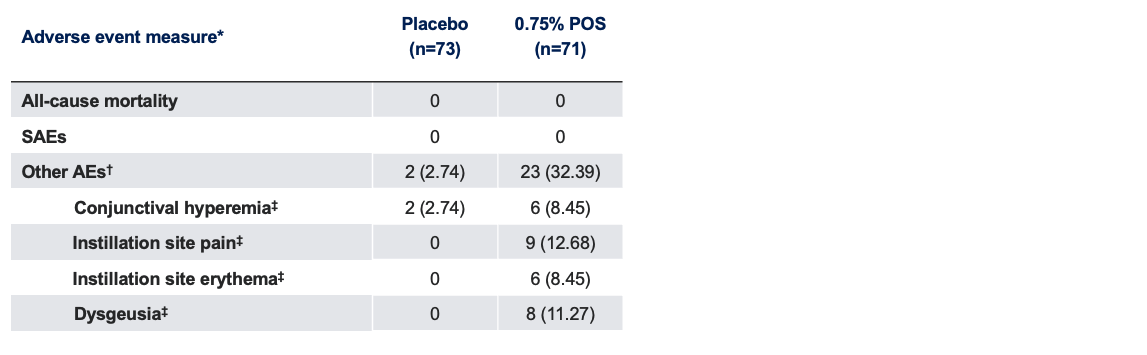
*Data presented as n (%); †5% frequency threshold for reporting other AEs; ‡Collected by systematic assessment.
AE, adverse event; POS, phentolamine ophthalmic solution; SAE, serious adverse event.
https://clinicaltrials.gov/study/NCT04638660.

